2 professionals
Bioderma Congress Reports AAD 2026
Bioderma Congress Reports AAD 2026
Get access to exclusive dermatological services to increase your professionnal knowledge: +500 pathology visuals, clinical cases, expert videos
Benefit from valuable features: audio listening, materials to be shared with your patients
Stay informed about the upcoming events and webinars, latest scientific publications and product innovations
Already have an account? login now
Reports written by Dr Ben Esdaile (Consultant Dermatologist, London, UK) and Dr Joël Claveau (Dermatologist, Quebec).

Related topics
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Konstantinos Liopyris (New-York), Michelle Tarbox (Texas), Luc Thomas (France) and Christian Navarrete-Dechent (Chile)
Speaker: Konstantinos Liopyris (New York)
He discussed melanoma specific features. He discussed specific features to help differentiate between invasive and in-situ melanomas. Predictors for invasive melanoma are atypical blue-white structures, shiny white lines and polymorphous vessels. Predictors for MMIS are flat lesions with atypical networks, multiple small hyperpigmented areas, angulated lines, irregular blotches and regression structures. He discussed the potential benefits of treating in-situ melanomas in just one step with a 5mm margin. He advised caution with streaks, inverse networks and irregular dots and globules as these can be seen in both invasive and in-situ melanomas.
Speaker: Michelle Tarbox (Texas)
She discussed psoriasis with regular uniform dotted vessels and white scale. Eczema with patchy dotted vessels and red background and yellow scale. LP has minimal scale and a violaceous background and Wickham’s striae. She showed chaotic glomerular vessels in Bowen’s. In seborrhoeic dermatitis there is more yellow crust and scale. In rosacea you get polygonal vessels in the telangiectatic variant. She also showed an example of red-sperm-like vessels in cutaneous lymphoma.
Speaker: Luc Thomas (France)
His three golden rules were to be very suspicious of single digit nail dystrophy, beware red tumours arising from the nail plate and long-standing warts could be SCC. With callus in the distal nail bed due to trauma, he showed a hyperkeratotic plug in the distal nail with no change in nail plate thickness. Pyogenic granuloma is located on the nail bed, and the key is looking at the free edge. Onychopapilloma are narrow and arises from the lunula and he advised looking at the free edge with thinning of the nail plate and a keratotic plug. Some can have splinter haemorrhages. Onychomatricoma comes from the proximal fold. He advised looking at the free edge for holes in plate. SCC are usually ill-defined with non-specific vessels. Sometimes onychopapilloma can actually be early SCC. Finally, he showed amelanotic melanoma with examples leading to destruction of the nail plate.
Speaker: Christian Navarrete-Dechent (Chile)
The real challenge are the flat facial lesions. For raised lesions the main differential are BCC v. naevus with in focus vessels being a useful clue for BCC. He also showed MAY globules as another useful clue and can be seen well with UV dermoscopy. You may also get a loss of adnexal openings with UV dermoscopy in BCCs. In flat lesions in naevus you get interfollicular pigmentation. In melanoma it is peri-follicular involvement. He showed an updated melanoma progression model with perifollicular projections prior to rhomboidal structures in the interfollicular skin in lentigo maligna. The session was completed by Kelly Nelson and Jennifer Stein with some interactive dermoscopy cases.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Kelly Nelson (Texas), Elizabeth Seiverling (Boston), Drew Emge (Kansas)
I started the conference bright and early at the Dermoscopy to the Rescue Session on skin cancer. Kelly Nelson from Texas discussed the importance of sharply-focused blood vessels in differentiating a nodular Basal Cell Carcinoma from nodular amelanotic melanoma. Desmoplastic melanoma can be difficult to detect especially in lentigo maligna. Using palpation can help detect induration and a possible invasive component. Be careful treating superficial components with topical imiquimod if a palpable component is present. She showed how dermoscopy can help in mapping tumours in challenging sites such as acral melanoma when working in a multi-disciplinary team.
Speaker: Elizabeth Seiverling (Boston)
She discussed important features including branching blood vessels, blue-grey ovoid nests and globules as well as leaf-like structures. She also discussed shiny white structures in tumours especially blotches and strands in basal cell carcinoma. Spoke wheels are very specific for basal cell carcinomas. Nodular basal cell carcinomas are more likely to have larger vessels and ovoid nests. She then showed examples how dermoscopy can help guide management of basal cell carcinomas with teledermoscopy cases. She also showed a case of MAY globules as a feature for higher risk micronodular basal cell carcinomas.
Speaker: Drew Emge (Kansas)
He discussed the progression model with the evolution of blood vessels and white structureless areas. In Actinic keratoses he discussed the strawberry pattern, pigment scale, hyperkeratotic follicles and background erythema as well as rosettes on polarised dermoscopy. In in-situ SCC the presence of dotted, glomerular vessels, erosions and scale are helpful. In early SCC looped vessels are a useful clue with the presence of white structureless areas and then some linear irregular vessels. As the tumour develops the keratin plugs develop in the follicle with yellow circles and ulceration with the development of a central keratin mass.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Ashfaq Marghoob (New York), Jason Lee (Philadelphia), Harold Rabinowitz (Miami), Dawn Hirokawa (Delaware) and Jennifer Stein (New York)
Speaker: Ashfaq Marghoob (New York)
Different structures relate to anatomical sites. He also discussed about negative networks and reticular depigmentation. The network is formed by the pigment in the rete ridges. The negative pigment network encompasses two main entities. The negative pigment network is formed by large pigment globules that are elongated-curvilinear globules consuming and coalescing in rete ridges. The reticular depigmentation is caused by white lines found in melanoma, spitz, SCC, dermatofibroma, clear cell acanthoma. Shiny white lines are a different entity only seen in polarised light. Angulated lines tend to be grey in colour and create polygons. These are formed by flattening of DE junction, confluence of melanocytes along DE junction and melanophages in the dermis. Streaks are radial projections outwards that represents the radial growth phase seen in superficial spreading melanoma and a reed naevus. Dots can appear as different colours and based on colour give clues to where they are located. Black dots represent melanin in stratum corneum that can be proven by tape stripping. Brown dots on the lines are seen in benign naevi. Brown dots in the network represents a nest in the dermal papillae. Brown dots not associated to the network can be seen in melanoma. Blue-grey dots represents free melanin or in melanophages can be seen in LPLKs or regressing melanoma. Globules can be different colours and white globules can be seen in balloon cell changes. Shiny white lines are only seen in PD are due to collagen disruption/stromal alteration. Blue-white veil is defined by a palpable area with a blue-white colour.
It corresponds to deep seated melanocytes with overlying orthokeratosis. Regression structures can also have a blue-white veil but are not palpable and are flat. They can present with scar-like depigmentation and granularity. A Blotch is large concentration of melanin that obscures underlying structures. Some can be removed by tape stripping (lamellar blotch). In basal cell carcinomas, leaf like structures and variants including spoke-wheel structures, concentric globules all represent tumour islands. Blue grey structures come as dots, globules or larger ovoid nests. The dots and globules represent tumour islands. MAY globules seen in both PD and NPD represent dystrophic calcification of tumour islands sometimes better seen in UV dermoscopy.
Speaker: Jason Lee (Philadelphia)
He discussed benign lesions and advised looking at the edge of haematomas under the nails or skin for clues for satellite blood spots. He showed examples of lacunae in haemangiomas, he also showed cases of dermatofibromas with multiple presentations including white scar-like area with delicate peripheral network. He also show examples of sebaceous induction on DFs on the shoulder with yellow globules. Seborrhoeic keratoses can be challenging in some cases especially in the presence of a network and some of the hyperpigmented variants. He moved on to discussing basal cell carcinomas and the useful clue of ulceration as well as the pigmented structures.
Speaker: Harold Rabinowitz (Miami)
He advised to beware grey colour in melanocytic lesions particularly on sun damaged skin. Malignant melanocytic lesions appear to have more asymmetrical dermoscopic patterns and multiple colours.
He discussed common naevus patterns and highlighted the importance of recognising these. He showed variants of naevi in age, skin colour and location. Reticular patterns are more common on the legs and so beware if you see a new globular pattern naevus in adults on the legs
Speaker: Dawn Hirokawa (Delaware)
She discussed the atypical pigment network representing architectural disorder. She showed atypical dots and globules that are asymmetrical in shape, size and location. She also showed examples of irregular blotches, shiny white structures, blue-white veils, negative pigment networks, angulated lines and irregular streaks seen in melanoma.
Speaker: Jennifer Stein (New York)
She discussed that histologically the anatomy is different. For facial dermoscopy she discussed the follicular openings leading to a pseudonetwork in pigmented lesions. In lentigo maligna you develop asymmetrical follicular openings (semi-circles) and perifollicular linear projections, grey dots (annular granular pattern) with curves eventually forming rhomboidal structures. She also discussed the absence of benign features. She then discussed the benign patterns on acral skin with parallel furrow, lattice-like and fibrillary patterns. She then highlighted the malignant parallel pattern or multicomponent pattern. Finally, she looked at nail unit melanoma and the challenge in visualising structures under the nail plate. The clue to melanocytic activation being a more grey colour. The true melanocytic proliferations either naevus or melanoma with clues to melanoma being atypical thickness of the bands and Hutchinson’s sign.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Antonella Tosti from Miami, Lidia Rudnicka from Poland, Natasha Mesinkovska from California, Daniel Melo from Brazil, Amy McMichael from North Carolina, Mariya Miteva from Miami, Rodney Sinclair from Australia, Rodrigo Pirmez from Brazil, Jerry Shapiro from New York, Isabella Doche from Brazil, Daniel Asz-Sigali from Mexico, Luis Sanchez from Mexico, Leopoldo Santos from Brazil, Elise Olsen from North Carolina, Antonella Tosti from Miami, Crystal Aguh from Baltimore, Leonard Sperling from Washington, Sergio Vano-Galvan from Spain, Rodrigo Primez from Brazil and Wenyo Wu from Shanghai
Speaker: Antonella Tosti (Miami)
There is still a debate as to whether hair loss is a side effect or a consequence of weight loss. There does seem to be more hair loss associated with greater weight loss. Mechanisms include protein deficiency because of a negative nitrogen balance. Hair loss is not always reversible so always do trichoscopy to evaluate the patient as they may have underlying AGA. In terms of management she suggests optimising protein intake. She advises topical steroids under occlusion for telogen effluvium. If AGA consider topical or oral minoxidil and consider 5-alpha reductase inhibitors/anti-androgens. There are potentially some benefits of GLP-1 in cases of CCCA in the presence of insulin resistance.
Speaker: Lidia Rudnicka (Poland)
Trichoscopy findings include perifollicular scaling with loss of follicular units and may be some perifollicular erythema (early sign with perifollicular arborising vessels). There is usually no scale between the hair follicles. In terms of management the first line is super potent topical steroid or IL steroid injections. In terms of oral therapy in moderate disease the first line is Hydroxychloroquine 200mg twice daily. The second line would be tetracyclines. In severe disease, consider methotrexate or Mycophenolate mofetil and then potentially JAK inhibitors. Cetririzine is a good option for itch associated with LPP (10mg daily). She also considers the addition of oral minoxidil starting at 1mg daily. For FFA her first line is finasteride 2.5mg-5mg daily and oral minoxidil and IL steroid injections. In some cases she adds isotretinoin, hydroxychloroquine or doxycycline.
Speaker: Natasha Mesinkovska (California)
Three drugs are currently available in the US -Baricitinib, Ritlecitinib and Deuruxolitinib. These drugs are meant to be used lifelong. Treat early and see every 3 months. Have little expectations in the first 3 months. At 6 months she would expect some response. Expect 20-30% to reach a SALT 20. She advises to be patient at least to 9 months. She would suggest only switching after 9-12 months if no response. Be cautious with JAKi switching but she advised to overlap for 2 months on alternate days.
Daniel Melo from Brazil gave a talk about Folliculitis decalvans and Dissecting Cellulitis. In terms of trichoscopy findings in Folliculitis decalvans he showed the classical polytrichia (tufting) and perifollicular scale as well as yellow crust and pustules. In management, his first line oral antibiotics are lymecycline as well as rifampicin and clindamycin.
Topical treatments include topical dapsone and steroids. Other options include oral isotretinoin. In dissecting cellulitis early disease can mimic alopecia areata. In terms of management first line therapy is oral isotretinoin. He also like using lymecycline with good responses in both diseases.
Speaker: Amy McMichael (North Carolina)
In terms of Discoid Lupus in skin of colour some trichoscopic features seen in white skin are not seen and can be more subtle. She discussed the Neopolitan trichoscopic pattern looking for those three colours. In terms of treatment she would suggest topical or intralesional corticosteroids and then hydroxychloroquine. In terms of traction alopecia sometimes the inflammation can only be seen by trichoscopy with the flambeau sign (resembling a torch) with numerous hair casts (a sign of active traction). Advise to make the hair styles looser and reduce friction. In terms of therapy, Topical steroids/ I/L steroids and topical minoxidil are first line. In CCCA, on trichoscopy there can be erythema and perifollicular hyperpigmentation and scale and grey-white halos. In terms of treatment talk about genetic predisposition and then treat the inflammation and fibrosis.
Speaker: Mariya Miteva (Miami)
She discussed anagen inhibition and showed trichoscopy is useful in making the diagnosis and for monitoring. There are 3 phases – acute phase, the subacute patch and then the chronic stage. Broken and exclamation mark hairs as well as yellow dots are useful clues. Black dots are cadaverised hairs. Yellow dots not specific but are helpful. Don’t expect to see yellow dots in young children or elderly patients as they don’t have the sebum. Circle hairs (pigtail hairs) are a type of short regrowing hair and another clue.
Speaker: Rodney Sinclair (Australia)
The primary goal is to stop further hair loss, the second was to stimulate hair growth and the third was to improve the quality of life of the patients. Patients were split into 3 groups - mild (SALT 20), Moderate (SALT 20-50) and severe (SALT 80). In under 12s the first line should be topical steroids with off label use of topical or oral minoxidil. For moderate disease the second line treatment would be JAKi or intralesional steroids. Similar recommendations in adults with intralesional steroids with the addition of oral minoxidil as an adjunctive treatment. If someone has episode of less than 6 months there is a high response rate to systemic steroids with a chance of coming off the drug (in about 40%).
Speaker: Rodrigo Pirmez (Brazil)
The main clue to look for in trichoscopy is hair follicle miniaturisation with hair shaft diameter diversity (greater than 20%). As it progresses you get more thin hairs. He suggested comparing with occipital scalp as most of the clues are at the very front of the scalp. In advanced AGA lots of single hair units and empty hair follicle with time develop (yellow dots). Some challenging cases where AGA can be misdiagnosed are in the presence of white hair roots, in the presence of perifollicular scaling consider FAPD (Fibrosing Alopecia in a Pattern Distribution) and consider a scalp biopsy, in CCCA you will find perifollicular scaling and white halos and in frontal fibrosing alopecia there will be a loss of vellus hairs being an important clue.
Finally, he discussed differentiating chronic Telogen Effluvium (CTE) and AGA. In AGA there is the presence of hair follicle miniaturisation and hair shaft diameter variability. In AGA there also tends to be a history of gradual loss where as CTE there is a history of shedding. In CTE there also tends to be varying hair lengths leading to an uneven hair length. In CTE the parting is normal and there is often temporal thinning as a hallmark. Sometimes multiple short re-growing hairs can be seen in CTE.
Speaker: Jerry Shapiro (New York)
In adult females he showed a study showing Spironolactone and oral minoxidil with good results. There are new studies looking at sublingual minoxidil in men with the aim of avoiding first pass metabolism and potentially less side effects. There are also studies for extended release oral minoxidil with encouraging phase II clinical trials. He showed data that hair tends to stabilises after 12 months on minoxidil and then hair diameter tends to slowly decline. There is some evidence that it is better to take minoxidil in the morning due to the circadian rhythm.
Speaker: Isabella Doche (Brazil)
She discussed topical finasteride as a possible option with varying results in clinical trials. Some emerging data with dutasteride but it is a much larger molecule and is still in the experimental stage. There are some promising topical anti-androgens such as clascoterone with comparable data to minoxidil. For injectables, mesotherapy results are mixed with low quality studies with the most studied being injectable dutasteride.
Speaker : Daniel Asz-Sigali (Mexico)
There are multiple filler types with the commonest being Hyaluronic acid. There are two main pathways causing filler induced alopecia. The first leads to extravascular pressure tamponade of the hair follicle (pressure alopecia) and the second is a more acute presentation with embolic damage to the hair follicle resulting from vascular occlusion from the filler. In terms of management immediate hyaluronidase and an ultrasound to see if any vascular occlusion is essential.
Speaker : Luis Sanchez (Mexico)
He showed it can be helpful for mapping biopsies in scarring alopecia as well as helping in a number of inflammatory scalp conditions as well as hair shaft disorders.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Boni Elweski (Alabama), Adam Rubin (New York), Phoebe Rich (Oregon), Tracey Vlahovic (California), Angela Jiang (Oregon), Mark Holzberg (Georgia), Jackson Turbeville (Wisconsin), Nathanial Jellinek (Rhode Island) and Julia Baltz (Rhode Island)
Speaker: Boni Elweski (Alabama)
In terms of bedside diagnosis, she gave tips for making the diagnosis of DLSO. Her tips were that abnormal fingernails are usually psoriasis and that asymmetrical nail dystrophy, the presence of tinea pedis and dermatophytoma (yellow or orange patches) point toward onychomycosis. In terms of oral options, she suggested oral terbinafine (250mg daily), oral fluconazole (200-400mg once weekly or twice weekly) and either pulsed or constant oral itraconazole (200mg 2 x day pulsed for 6-8 months). In terms of monitoring – consider checking LFTs before and 3 monthly. In terms of itraconazole there are interactions particularly if taking a statin and patients should avoid antacids when taking the medications. There have been case reports of heart failure with itraconazole.
Speaker: Adam Rubin (New York)
He discussed the wide differential of solitary erythronychia with the commonest cause being onychopapilloma but other benign and malignant lesions are in the differential. A longitudinal biopsy is needed in some cases to confirm the diagnosis as there are malignant variants.
Speaker: Phoebe Rich (Oregon)
She talked about acute, chronic and other paronychias. Acute paronychias last less than 6 weeks and are usually staph or strep related. She discussed TB as a cause of painless paronychia.
In terms of management of mild acute paronychia then warm water soaks +/- vinegar or iodine. If inflamed with pus then topical or oral antibiotics. Chronic paronychia lasts longer than 6 weeks and is a chronic inflammatory dermatitis often related to chemicals and water. The main management is avoiding irritants. Squamous cell carcinoma can mimic paronychia in the chronic setting.
Speaker: Tracey Vlahovic (California)
She talked about nail devices but these do not tend to lead to long term improvement. She showed the use of Tazarotene 0.1% gel as an adjunctive therapy with some benefit. She talked about a suture technique by leaving a suture in the nail for 6 weeks. She suggested making sure there is no underlying nail deformity.
Speaker: Angela Jiang (Oregon)
She described that this usually is caused from damage to the onychodermal band. There are a number of causes including environmental as well as psoriasis, onychomycosis, systemic causes and drugs. Clues to an environmental cause are the presence of irritancy and onychoschizia. Dermoscopy can be helpful in differentiating onychomycosis (jagged/spiked proximal edge) compared to smooth edges for traumatic onycholysis. In terms of general care, she suggested clipping the affected nails back, minimise hand washing and refrain from nail cosmetics. Co-infection is common including pseudomonas and fungal infection. She discussed the use of tretinoin gel for 3 months with some cases of benefit. It appears to work by delaying keratinisation and helping nail plate adherence. She also discussed allergic contact dermatitis as a cause or onycholysis.
Speaker: Mark Holzberg (Georgia)
Treatments can lead to disfigurement. He highlighted that some will need a biopsy to exclude squamous cell carcinoma. In terms of options there are a few options. For isolated warts on the skin then destructive methods such as liquid nitrogen is first line. If periungual then immunotherapy with a contact allergen, bleomycin or cidofovir. He discussed the possible role of oral zinc to boost cell-mediated immunity as well as immunotherapy with the HPV vaccine.
Speaker: Jackson Turbeville (Wisconsin)
He discussed nail psoriasis in terms of management first line with high potency corticosteroids +/- calcipitriol, Systemics include methotrexate, biologics and apremilast. In nail lichen planus he discussed the risk of permanent scarring. Prompt diagnosis is essential and management with either intralesional or intramuscular steroids are first line. Second line treatments include oral acitretin and methotrexate. There is emerging evidence for low dose naltrexone 3mg daily. He discussed physical disorders including congenital hypertrophy of the lateral nail fold. Leukonychia punctata is usually related to trauma and not nutritional deficiencies. He concluded with habit-related disorders with onychophagia, onychotillomania and discussed N-acetylcysteine as an emerging pharmacotherapy.
Speaker: Nathanial Jellinek (Rhode Island)
He discussed the importance of managing the patient and of good anaesthesia. He showed techniques for distal nail blocks and discussed ways to stop bleeding with pressure and the new role of tranexamic acid.
Speaker: Julia Baltz (Rhode Island)
In terms of assessment, she suggested splitting this into monodactylous or polydactylous. She showed onychomycosis as a potential cause with the reverse triangle sign on dermoscopy. For monodactylous she further split this into melanocytic and non-melanocytic causes. She showed that melanonychia in childhood is almost always benign, but you need to be much more suspicious in adulthood.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Cristian Navarrete-Dechent (Chile), Nadeem Marghoob (Boston) and Ashfaq Marghoob (New York)
Speaker: Cristian Navarrete-Dechent (Chile)
These can be very challenging especially flat facial lesions. The facial anatomy is different with flat skin with interruption just from adnexal openings forming a pseudonetwork.
The junctional naevus is less common with pigmentation between the adnexal opening. In melanoma there is adnexal accentuation. He showed the dermoscopic features of solar lentigo with curved parallel lines, moth-eaten borders and reticular patterns with symmetrical follicular pigment. In early seborrhoeic keratoses classical features appear. In pigmented lichen planus-like keratosis he showed cases of coarse granules/peppering. He advised caution if the only feature is fine granularity. In pigmented actinic keratoses clues are pigmented scale, strawberry pattern, rosettes, dilated follicles and annular-granular pattern. In lentigo maligna the diagnosis can be challenging. He discussed the updated facial melanoma progression model. He discussed it starts with asymmetrical follicular pigmentation followed by perifollicular linear projections (PLPs). When the PLPs curve around the follicle this gives the circle with circle pattern seen in Lentigo maligna. They then start forming rhomboidal structures and eventually lead to follicular obliteration in invasive lentigo maligna melanoma. He then highlighted the importance of using the clinical appearance and palpation as well as dermoscopy to help make the diagnosis. He showed that in recurrent melanoma the most common pattern will be homogenous pigmentation as the scar will obliterate the follicular openings.
Speaker: Nadeem Marghoob (Boston)
He discussed the three main benign patterns – parallel furrow pattern, lattice pattern and fibrillar pattern. He advised to be cautious if you see a fibrillar pattern on the palms. In terms of malignant patterns he discussed the parallel ridge pattern and multi-component patterns as the 2 commonest patterns of acral melanoma. He showed benign volar melanotic macules in skin of colour with a parallel ridge pattern. If acral naevi are homogenous, reticular, globular or non-typical consider monitoring.
Speaker: Ashfaq Marghoob (New York)
He showed how to differentiate blood and the danger of an underlying melanoma. He suggested looking at the distal nail plate for the present of pigment. He showed the jagged proximal edge as well as an inverse triangle sign in onychomycosis. If the band arises from the lunula the free edge can be helpful to see where in the matrix the band arises. Onychopapillomas and SCC look the same in early disease. If multiple onychopapillomas consider BAP1 syndrome. For melanocytic lesions think of melanocytic activation versus proliferation. The free edge helps decipher whether the lesion originates from the distal or proximal nail plate. He highlighted four factors that eclipse dermoscopy for risk of acral melanoma- band greater than 6mm, Hutchinson’s sign, triangular shape and nail dystrophy. In terms of activation (normal melanocytes producing melanin) they tend to be grey or light tan that is more homogenous. In terms of naevus or melanoma – naevi tend to be organised whereas melanoma tends to be disorganised. Finally, he highlighted that congenital naevi in children break all the rules and can look extremely concerning.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Robert Novoa (California), Shanni Wongvibulsin (Los Angeles), Zhu Ran Cai (Montreal), Shruti Agrawal (Minnesota), Jenna Lester (San Francisco), Roxana Daneshjou (Boston), Tofunmi Omiye (California), Victoria Mar (Melbourne) and Justin Ko (Boston).
Speaker: Robert Novoa (California)
He discussed supervised learning with ground truth answers, unsupervised learning where we don’t have the ground truth and reinforcement learning where the algorithm is rewarded rather than ground truth answers. Large Language models are being trained on the entire internet. He discussed the amazing improvement in generative AI in the last few years.
Speaker: Shanni Wongvibulsin (Los Angeles)
She showed examples of Generative AI to develop presentations. She showed Open Evidence and using Dot Flows to do patient leaflets or insurance claims. She showed how AI scribes can help prevent burnout and improve health practitioner well-being. She showed how Notebook LM can help in medical education in creating notes, presentations and podcasts. AI tools can also be used for developing detailed research plans. She discussed AI doctors and the potential risks of hacking and false advice given by some tools.
Speaker: Zhu Ran Cai (Montreal)
He discussed this in four main areas. He highlighted the illusion of reasoning with algorithms with subtle changes to data leading to poorer outcomes. He showed how some LLMs don’t know what they don’t know and were overconfident. He highlighted the danger of hallucinations in LLM with examples of errors in radiology and pharmacology. The final area he covered was the patient-AI hazard. He showed that 230million people ask ChatGPT medical questions every week with many receiving incorrect advice. He concluded that AI still needs to prove its worth in the real-world setting.
Speaker: Shruti Agrawal (Minnesota)
She showed an algorithm for detecting naevoid melanoma and melanoma lymph node metastases. She discussed the role of AI in triaging slides to aid the Dermatopathologist. She did mention this runs the risk of diagnostic bias. She discussed the challenge of no ground truth in some cases.
Speaker: Jenna Lester (San Francisco)
She discussed the lack of transparency of the data sets that have trained many of the algorithms available. She showed the lack of diversity in skin tones in data sets. She showed that improving the diversity of data sets actually appears to improve the overall performance of the algorithms on all skin tones. She also raised the point that community engagement is an essential part of building these datasets.
Speaker: Roxana Daneshjou (Boston)
She believes that in the next 10 years, the entire field of medicine will undergo major upheaval. She believes that patients will turn to consumer models due to lack of access to care.
Speaker: Tofunmni Omiye (California)
He talked about Agentic AI - Autonomous AI working without oversight. He discussed how exciting the field is but concerns over error handling given the amount of autonomy.
Speaker: Victoria Mar (Melbourne)
They are developing AI to help triage the higher risk patients and reduce unnecessary biopsies.
Speaker: Justin Ko (Boston)
He feels that humans may surrender their judgment as they are so convinced by AI outputs. He talked about potential harms with the use of AI. There are important benefits with eye-to-eye contact with patients with ambient AI devices. This will lead to a reduction in administrative burden and improve quality of doctor-patient interactions.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Seemal Desai (Dallas), James Del Rosso (Las Vegas), April Armstrong (California), Linda Gold (Detroit), Melanie Palm (San Diego), Jerry Shapiro (New York) and Darrel Rigel (New York).
Speaker: Seemal Desai (Dallas)
For Atopic dermatitis he showed some 7-year safety data for upadacitinib with no new safety signals. In Alopecia Areata there are now 3 FDA approved treatments. In psoriasis he discussed a new TYK-2 inhibitor (Envudeucitinib) with impressive results. In hand eczema Delgocitinib, a topical pan JAK inhibitor, shows some excellent results. In vitiligo he showed some breaking results of oral upadacitinib but warned this will take long periods of treatment to get results.
Speaker: James Del Rosso (Las Vegas)
In topical therapies we now have agents for treating all four pillars of acne: excess sebum (clascoterone), follicular hyperkeratinisation (retinoids), cutibacterium (BPO and antibiotics) and inflammation (dapsone and azelaic acid). Clascoterone is better twice a day rather than once daily and also takes time to work. He also discussed Sarecycline as a narrower spectrum antibiotic for acne.
Speaker: April Armstrong (California)
For atopic dermatitis she discussed topical PDE-inhibitors and JAK inhibitors. Roflumilast 0.05% cream for children aged 2-5 years of age and Ruxolitinib 1.5% cream can be used twice daily in the paediatric population. A new drug Difamilast 1% ointment is a selective PDE4 inhibitor that is used 2 x daily with impressive results in clinical trials. Tapinarof 1% cream is approved for children 2 years and above.
In hand eczema Delgocitinib, a topical pan JAK inhibitor, shows some excellent results. For psoriasis Roflumilast topical foam 0.3% is FDA approved for the scalp and body in ages 12 and over.
Speaker: Linda Gold (Detroit)
She discussed the topical PDE4 inhibitors, aryl hydrocarbon receptor agonists and topical JAK inhibitors. In oral therapy she showed good safety data with abrocitinib and upadacitinib. She then discussed new STAT 6 targets being developed.
Speaker: Melanie Palm (San Diego)
She discussed the problems with deeply delivered devices that can affect fibrosis and plane dissection during skin surgery or face lifts. She then discussed the role of tranexamic acid in MOHS surgery at reducing blood loss post operatively when TXA was given topically or by injection.
Speaker: Jerry Shapiro (New York)
He talked about a non-ablative fractional laser (1565-nm) to regrow hair with some promising results. Exosomes are popular in New York and show some good results but are not FDA approved. There is now a new form of extended-release minoxidil (VFPHL01) with encouraging Phase II clinical results. Clascoterone 5% solution is going to be used in the scalp and the preliminary results are impressive in trial data. He also discussed some new topical preparations to stimulate hair follicle stem cells.
Speaker: Darrel Rigel (New York)
He discussed a larger study into nicotinamide for chemoprophylaxis that showed that the earlier you start the more the effect. He discussed ways to identify the higher risk SCC patients. He showed good results of curettage followed by imiquimod for superficial BCCs. He discussed a new polyomavirus antibody test for following up Merkel Cell carcinoma. In terms of melanoma diagnostics he talked about the rising AI devices in melanoma detection.
Report written by Dr Ben Esdaile (Consultant Dermatologist, London, UK)
Speakers: Jerry Shapiro (New York), Natasha Mesinkovska (California), Leopoldo Santos (Brazil), Lidia Rudnicka (Poland), Elise Olsen (North Carolina), Antonella Tosti (Miami), Crystal Aguh (Baltimore), Leonard Sperling (Washington), Sergio Vano-Galvan (Spain), Rodrigo Pirmez (Brazil) and Wenyo Wu (Shanghai)
Speaker: Jerry Shapiro (New York)
In terms of treatment he described about 70% success rate in stopping the disease and this may take around 10 months. If rapidly progressive, then oral prednisolone is his preferred option. If slowly progressive, he uses a combination of topical tacrolimus/minoxidil 5% and clobetasol. He also discussed intralesional steroids. If these fail then other options include low dose doxycycline, hydroxychloroquine and dutasteride (in post-menopausal women) or finasteride in younger women. For facial papules in FFA he recommends isotretinoin with a loading dose of 40mg/day for 2 weeks then 10-20mg 3 x week for 3 -6 months. He tells patients to avoid chemical sunscreens and avoid fragrance.
Speaker: Natasha Mesinkovska (California)
She advised to treat early. In terms of JAK inhibitors, she discussed Ritlecitinib and the fact that JAKs work and appear safe. The latest JAK is Deuroxolitinib 8mg twice daily. Results show they can achieve a SALT 20 in just 8 weeks. JAKs are for long term treatment and appear safe. She discussed some risk of HSV. She discussed monitoring including FBC, Liver function tests and lipid profiles. She also discussed the contraindication if malignancy, significant cardiac history or previous venous thromboembolism. Finally, she discussed the role of dupilumab and tralokinumab in AA patients with atopic eczema.
Speaker: Leopoldo Santos (Brazil)
He discussed 5-alpha reductase inhibitors for mesotherapy and showed that dutasteride appears to be a better choice over finasteride and requires re-injection every 3 months rather than weekly or bi-weekly. There may however be a systemic reduction in DHT and potential side effects. He advises mesotherapy should be added to oral treatment. He discussed the use of Bicalutamide (androgen receptor antagonist) in mesotherapy but with limited improvement and it can be painful.
Speaker: Lidia Rudnicka (Poland)
She gave tips on how to differentiate AGA from other hair loss disorders. She highlighted the dermoscopic features of AGA with miniaturisation of the hair shafts resulting in hair shaft thickness heterogenicity, multiple units with one hair and the peripilar sign (perifollicular hyperpigmentation). She showed the presence of vellus hairs in helping differentiate AGA from FFA.
Speaker: Elise Olsen (North Carolina)
She showed that topical minoxidil leads to very little systemic absorption. She showed not much difference in topical minoxidil versus oral finasteride. Doses of 5-10mg can cause more potential side effects. If cardiac side effects, consider splitting the dose and increase fluid intake. Pericardial effusions of significance are rare and would present with pleuritic chest pain and SOB on exertion. 10% can experience hair shedding and up to 14% can get headaches. Concomitant spironolactone can be useful in FPHL for any hirsutism and potentially ankle swelling.
There are some potential new types of oral minoxidil including a sublingual formulation and an extended release oral minoxidil.
Speaker: Antonella Tosti (Miami)
With age, hair density as well as sebum production and pigmentation declines. The fibres are coarse and diameter reduces. Reversal of hair greying is possible, but the mechanisms are not fully understood. Scalp skin health also has an effect on the quality of hairs.
Speaker: Crystal Aguh (Baltimore)
There does seem to be a link with insulin resistance. If a personal of family history of diabetes, then consider the addition of oral metformin. If concomitant AGA consider addition of oral minoxidil. Specifically, for the CCCA, consider intralesional steroids, super potent topical steroids. Consider doxycycline especially with scalp pain. Consider oral antihistamines with itch. She describes CCCA as a fibroproliferative disorders and advised us to consider drugs that treat inflammation as well as fibrosis. She showed potential benefits of topical metformin, and she is now combining 10% metformin with a potent topical steroid.
Speaker: Leonard Sperling (Washington)
He discussed choosing the correct site and the importance of good clinical information and the consideration of the addition of a clinical image. For non-cicatricial alopecias aim for the most involved site but for cicatricial alopecias aim for almost the opposite to get early involvement.
Speaker: Sergio Vano-Galvan (Spain)
He showed a large study in his unit supported its safety with discontinuation in only 1.7% with the most common side effects being hypertrichosis, leg oedema and facial oedema and tachycardia. In patients over 65 or older the side effects were very slightly higher with a withdrawal rate of 2.8% with the commonest cause being tachycardia. He concluded that LDOM is safe to use in hypertension without referral to a cardiologist but to be cautious if a patient is taking doxazosin or 3 or more anti-hypertensive drugs.
Speaker: Rodrigo Primez (Brazil)
It has features of both AGA and FFA. Trichoscopy shows variability in hair shaft diameter but with follicular scaling and tufts of hair. Many patients present with itch, pain and burning. In terms of treatment for men, he suggested oral finasteride and oral minoxidil. In women, oral minoxidil as well as androgen receptor antagonists and 5-alpha-reductase inhibitors. For the inflammatory component he suggested topical steroids and then drugs such as hydroxycholoroquine or doxycycline. He also occasionally uses intralesional steroid injections.
Speaker: Wenyo Wu (Shanghai)
He discussed the history of hair transplantation in China and the use of robotic hair transplantation and the use of AI in assessing scalps prior to transplantation. He discussed the pros and cons of FUE – follicular unit extraction (no linear scar) over the FUT – follicular unit transplantation (linear scar). He then showed the use of a robotic unit transplantation showing improved speed and accuracy.
Reports written by Dr Joël Claveau (Dermatologist, Quebec)
Speaker: Cristian Navarrete-Dechent (Santiago, Chile)
Wrong-site surgery is one of the most frequent errors in dermatologic surgery and one of the leading causes of medical malpractice claims, accounting for approximately 14% of complaints in this specialty. Several factors compound the risk of site misidentification:
Pre-operative clinical photography is the single most effective strategy for correct surgical site identification. A well-taken photograph leaves no ambiguity and saves time, cost, and frustration for both clinician and patient. The evidence is compelling:
Clinical photographs were present in 67% of referrals and anatomical diagrams in 53.9%; both were significantly associated with correct site identification.
Recommended photographic technique:
Patient self-photographs (selfies) are acceptable when professional photography is unavailable.
Timing: photograph the lesion before any biopsy; if a biopsy has already been performed and the site remains visible, photograph it immediately afterward. Multiple angles are advisable for complex anatomical locations. Lesions of the nose are notably more often incorrectly described in referral letters, warranting extra care.
A multicenter retrospective study examined the impact of anatomical documentation quality on surgical site identification across two institutions – Pontificia Universidad Católica de Chile (PUC) and Memorial Sloan Kettering Cancer Center (MSKCC) – and found major discrepancies in laterality documentation rates (J Eur Acad Dermatol Venereol 2023;37(1):e93):
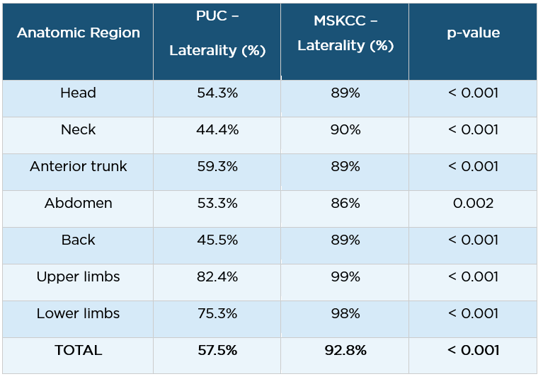
Overall, laterality was documented in only 57.5% of cases at PUC versus 92.8% at MSKCC (p < 0.001). This gap underscores that even basic anatomical documentation is far from universal, and that the presence of a clinical photograph is strongly correlated with higher overall referral quality.
When a pre-biopsy photograph is unavailable, several complementary tools can assist in locating the surgical site:
Standardized body diagrams using validated anatomical terminology (513 terms) provide a systematic approach to site description. The free online tool anatomymapper.com and the Delphi body map developed by the International Skin Imaging Collaboration (ISIC) offer standardized, reproducible documentation. Limitations include potential for subjective interpretation and challenges in transferring diagrams to electronic medical records.
After a partial biopsy, dermoscopy can help identify residual tumor tissue at the biopsy site:
After a complete excisional biopsy, the scar typically appears as a pink area with bright white (shiny white) structures under dermoscopy. Dermoscopy is rapid, widely available, and requires no additional equipment beyond what most dermatologists already use.
An emerging technique using a 365 nm ultraviolet light source with a UV filter to capture fluorescent emission from cutaneous chromophores. This modality is now available in commercial handheld dermoscopes, is easy to use, and is particularly helpful in detecting residual lesion material after a partial biopsy. It represents a promising adjunct that can be integrated into standard practice with minimal additional investment.
Confocal microscopy offers near-histological cellular resolution, enabling visualization of the epidermis and papillary dermis non-invasively. It is particularly valuable for identifying subtle residual lesions after a partial biopsy and for real-time follow-up monitoring. Its main limitations are high cost, limited availability (primarily in academic or specialized centers), and a steep learning curve.
Key Takeaways
Reports written by Dr Joël Claveau (Dermatologist, Quebec)
Speakers: Anna Cogen, MD, PhD, FAAD, Brittany L. Dulmage, MD, FAAD, Ashley Gray, MD, Rachel Kirven, MD, FAAD, Eric Mukherjee, MD, PhD, Connie Shi, MD, FAAD
Overview: Approach to Morbilliform Eruption
When evaluating a new rash, the first step is to confirm the morphology. Morbilliform eruptions that display high-risk features including trunk and facial involvement, facial or hand edema with the "oblique earlobe crease" sign, pustules, vesicles, duskiness, palmar/plantar involvement, or mucosal/genital involvement should raise concern for a Severe Cutaneous Adverse Reaction (SCAR). By contrast, low-risk reactions are trunk-predominant, spare the face, palms, soles, and mucosal surfaces, and lack pustules or vesicles. Bullous, vesicular, pustular, and purpuric morphologies should each trigger further SCAR workup including labs, biopsy, and diagnostic algorithms. Morphologies that do not fit these categories can exit the algorithm. The three main SCARs under the morbilliform spectrum are Acute Generalized Exanthematous Pustulosis (AGEP), Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), and Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis (SJS/TEN). Graft-versus-host disease and viral eruptions should also be considered in the differential.
AGEP (Acute Generalized Exanthematous Pustulosis)
AGEP is characterized by a rapid onset of 24–48 hours after drug exposure, though latency may be prolonged with beta-lactams, hydroxychloroquine, proton pump inhibitors, and terbinafine. Clinically, lesions are superficial, non-follicular pustules classically described as "lakes of pus," preferentially localized to intertriginous areas; palmoplantar involvement is rare. As the eruption evolves, collarettes of scale with superficial exfoliation develop. Mucosal involvement occurs in fewer than 20% of cases. Systemic features include fever and leukocytosis with neutrophilia. AGEP is more commonly observed in obese patients. Diagnosis is formalized using the EuroSCAR validation score, which assigns points across four domains: morphology (pustules, erythema, distribution/pattern, post-pustular desquamation), course (mucosal involvement, acute onset within 10 days, resolution within 15 days, fever ≥38°C, neutrophilia), and histology (exocytosis of neutrophils scores, subcorneal/intraepidermal pustules, spongiform pustules with papillary edema).
DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
DRESS has a characteristic latency of 2–8 weeks from drug exposure, though shorter latencies under 15 days have been reported with beta-lactam antibiotics and radiocontrast agents. The cutaneous pattern begins as a diffuse morbilliform eruption that may evolve into a polymorphic presentation including eczematous, vesiculobullous, pustular, erythrodermic, or purpuric (predominantly on the lower extremities) morphologies. Facial and ear edema is a hallmark feature, and the oblique ear crease sign is a clinically useful indicator. Mucosal involvement, when present, is usually mild or absent. Early symptoms relative to the onset of erythema include pruritus and pain, followed by fever, dysphagia, and lymphadenopathy. Lab findings include peripheral eosinophilia, atypical lymphocytes, early leukopenia transitioning to leukocytosis, and cytopenias in older patients. Systemic organ involvement is common, with hepatic and renal injury being most frequent.
Diagnosis is assessed using the RegiSCAR score, incorporating fever ≥38.5°C, lymphadenopathy, eosinophilia, atypical lymphocytes, skin rash extent beyond 50% BSA, rash suggestive of DRESS, skin biopsy findings, organ involvement, resolution ≥15 days, and exclusion of alternative causes by at least 3 negative tests (HAV/HBV/HCV, Mycoplasma, ANA, blood culture, Chlamydia).
SJS/TEN (Stevens-Johnson Syndrome / Toxic Epidermal Necrolysis)
SJS/TEN typically develops 5–21 days after drug initiation. The cutaneous morphology is characterized by dusky or two-toned erythema, atypical targetoid lesions, and vesiculobullae. Distribution begins on the face and trunk early and extends to the palms and soles, with accentuation in pressure-related areas. Mucosal involvement is common and severe, distinguishing SJS/TEN from the other SCARs. Systemic findings include fever, anemia, and leukopenia (more common with high body surface area involvement). Key clinical clues include skin pain preceding blister formation and positive Nikolsky and Asboe-Hansen signs. Distinction between SJS (<10% BSA) and TEN (>30% BSA) carries prognostic significance.
Overlap Syndromes in SCARs
Early SCARs frequently display overlapping features, though most cases ultimately evolve into a single diagnosis. True overlap defined as meeting criteria for two or more SCARs simultaneously is observed in 2–3% of patients in large DRESS cohorts and is clinically important to recognize. The management principle in overlap scenarios is to prioritize the highest-risk SCAR phenotype: SJS/TEN + AGEP overlap should be treated as SJS/TEN; DRESS + AGEP overlap should be treated as DRESS; DRESS + SJS/TEN overlap requires individualized management guided by predominant skin severity (epidermal detachment, mucosal involvement) and systemic severity (extent of organ dysfunction).
Here is a useful concise comparison of the different SCARs entities to conclude:
Immunotherapy Background: Mechanism of Action
Immune checkpoint inhibitors (ICIs) work by unleashing cytotoxic T-cell activity against tumor cells. Under normal physiologic conditions, CTLA-4 on T cells competes with CD28 for binding to B7 on antigen-presenting cells, thereby dampening T-cell activation. Separately, PD-1 expressed on cytotoxic T lymphocytes binds PD-L1 on tumor cells, suppressing their anti-tumor activity. ICIs block these inhibitory pathways. The resulting immune activation drives efficacy but also produces immune-related adverse events (irAEs), of which cutaneous toxicities are the most common.
Maculopapular Rash: The Most Common Cutaneous irAE
Maculopapular or morbiliform rash is the most frequent cutaneous irAE, typically appearing 3–6 weeks after the initial ICI dose. Clinically, it presents as multiple erythematous macules and papules with minimal scale that may coalesce into plaques, predominantly distributed on the trunk and extensor surfaces of the extremities, with the face generally spared.
Symptoms may range from pruritic to entirely asymptomatic. Diagnosis is most often clinical, based on the timing and morphology, though skin biopsy may be performed when the presentation is atypical. On histology, the most common findings are vacuolar degeneration at the dermal-epidermal junction, perivascular lymphocytic infiltrate, and eosinophils; less commonly, epidermal spongiosis and necrotic keratinocytes are seen. Importantly, a maculopapular-appearing rash may represent the initial manifestation of other cutaneous irAEs including lichenoid dermatitis, eczematous dermatitis, psoriasiform dermatitis, or bullous pemphigoid. Biopsy is more strongly indicated when the rash presents more than 2 months after ICI initiation, involves mucosal or acral surfaces, is associated with skin pain or severe pruritus, or when the patient has a personal or family history of psoriasis, eczema, or lichen planus.
Management of Maculopapular irAE: Grading & Treatment
Management is guided by the Common Terminology Criteria for Adverse Events (CTCAE) grading system. Grade 1 (macules/papules <10% BSA, with or without symptoms) can be managed while continuing immunotherapy, using topical emollients, oral antihistamines for pruritus, and moderate-potency topical steroids. Grade 2 (10–30% BSA, limiting instrumental activities of daily living) similarly permits continuation of ICI, with escalation to moderate-to-high potency topical steroids with consideration of prednisone 0.5 mg/kg/day if unresponsive to topicals. Grade 3 (>30% BSA, limiting self-care activities of daily living) mandates holding immunotherapy, initiating high-potency topical steroids, and starting systemic prednisone at 0.5–1 mg/kg/day. Biopsy or inpatient care should be considered at this stage. For other specific irAE subtypes, management differs based on the etiology.
Lichenoid dermatitis is treated with topical steroids, calcipotriene, calcineurin inhibitors, NB-UVB, hydroxychloroquine, or acitretin while eczematous dermatitis adds dupilumab to the same topical regimen.
Psoriasiform dermatitis may benefit from ustekinumab, apremilast, or methotrexate while bullous pemphigoid warrants high-potency topical steroids (or prednisone 1 mg/kg/day for severe disease), dupilumab, omalizumab, or rituximab.
Immunotherapy-Related SJS/TEN
The pathogenesis of SJS/TEN in the setting of immune checkpoint inhibition remains debated. A key case series by Molina et al. (JAAD 2020) showed that all patients had recently initiated concomitant medications known to cause SJS/TEN, including trimethoprim-sulfamethoxazole and allopurinol. These patients lacked ocular involvement and had a generally favorable treatment response. This supports a "two-hit" hypothesis in which ICI primes and amplifies cytotoxic T-cell responses, but a second drug exposure is required to trigger SJS/TEN. This showed that a thorough medication review for co-culprit drugs is essential.
Morbilliform Eruption After Allo-HSCT: Acute Cutaneous GVHD
After allogeneic hematopoietic stem cell transplantation (allo-HSCT), morbilliform eruption can represent acute GVHD, drug eruption, viral exanthem, or engraftment syndrome. Strict time-based definitions of acute GVHD (<100 days post-transplant) are no longer used. The allo-HSCT timeline begins with donor search and HLA testing, followed by pre-transplantation conditioning, infusion day, the early recovery period within the first month representing the highest-risk window for infections and vaso-occlusive disease when engraftment occurs. We usually consider the acute GVHD risk period to be the first 3 months post-graft, and the chronic GVHD period beyond 3 months. Engraftment is defined as ANC >500/mm³ for 3 consecutive days, occurring at approximately 10–14 days for autologous and 2–4 weeks for allogeneic transplants. Acute cutaneous GVHD most commonly presents as a morbilliform eruption although severe cases can progress to erythroderma, bullae, erosions, and desquamation resembling SJS/TEN (Stage IV).
Characteristic features include a folliculocentric appearance and acral involvement (palms, soles, dorsal hands/feet, ears, scalp). This must be distinguished from toxic erythema of chemotherapy/hand-foot syndrome caused by conditioning regimens. Other organ involvement like hepatic (transaminitis/cholestasis) and gastrointestinal (diarrhea) is characteristic of acute GVHD and helps confirm the diagnosis. Histopathology is often non-specific. Key risk factors for GVHD include HLA mismatch (most important), unrelated donor, older patient or donor age, female donor/male recipient, total body irradiation, and post-transplant events such as donor lymphocyte infusion and tapering of immunosuppression. Advancements in conditioning regimens (cyclophosphamide, abatacept) have led to a decrease in acute GVHD incidence.
Differentiating Acute GVHD from Drug Hypersensitivity & Engraftment Syndrome
Clinical differentiation between acute cutaneous GVHD and drug hypersensitivity reactions after transplant is aided by specific features. Facial involvement including the pinna is significantly more common in GVHD than in drug reactions, and combined facial plus palmar/plantar involvement was seen exclusively in the GVHD group. Diarrhea and hyperbilirubinemia were likewise observed only in GVHD patients, not in drug reaction patients. Engraftment syndrome represents a distinct entity where systemic findings arise during the engraftment period, most commonly after autologous HSCT. Diagnostic criteria include non-infectious fever, skin rash, pulmonary edema, weight gain, hepatic/renal dysfunction, and transient encephalopathy. Key features distinguish it from acute GVHD: engraftment syndrome occurs in both autologous and allogeneic transplants, peaks at 10–12 days (coinciding with myeloid recovery), diarrhea is absent, pulmonary edema and weight gain occur, and there is a rapid and robust response to systemic corticosteroids. Engraftment syndrome is often self-limited. Systemic steroids should be initiated if it is sustained beyond days.
Reports written by Dr Joël Claveau (Dermatologist, Quebec)
Speakers: Suephy C. Chen, Elizabeth Gates Berry, Caroline C. Kim
The nomenclature surrounding dysplastic nevi has been a source of clinical confusion for nearly two centuries. Key milestones include:
The presenters favor the term “atypical nevus” over “dysplastic nevus” to reduce confusion among patients and clinicians alike.
Atypical (dysplastic) nevi present a diagnostic challenge for several reasons:
A critical conceptual point: clinical atypia (defined by ABCE criteria) cannot reliably predict histologic dysplasia. There is a recognized “diagnostic gap” between the two. Dysplasia is further characterized using the “Brick Wall” analogy:
A common misconception is that atypical nevi are direct melanoma precursors requiring prophylactic removal.
The evidence does not support this:
Atypical nevi should be regarded primarily as markers of increased melanoma risk rather than obligate precursor lesions.
Dermoscopy is an essential tool for the evaluation of pigmented lesions. It improves diagnostic accuracy and helps discriminate between atypical nevi and melanoma, reducing the rate of unnecessary biopsies when used by trained clinicians.
Sequential digital dermoscopy imaging (SDDI) and total body photography are valuable adjuncts, particularly for high-risk patients. These tools allow for monitoring of change over time and support risk stratification without requiring immediate biopsy.
When biopsy is indicated (i.e., for lesions suspicious of melanoma), technique matters:
Important pitfall: A partial biopsy of a dysplastic/atypical nevus may create a “pseudo-melanoma” artifact, where regenerative changes in the scar tissue histologically mimic melanoma. This underscores the importance of complete excision when biopsy is performed.
Histologic evaluation of atypical nevi should be performed by an experienced pathologist, ideally a dermatopathologist. The WHO two-tier grading system (low-grade vs. high-grade dysplasia) is now recommended. Key histologic features assessed include architectural disorder and cytologic atypia, which may occur independently or in combination.
The reproducibility of pathological grading of dysplastic nevi remains a significant challenge. Even among experts, inter-observer agreement is imperfect, and this must be recognized when making clinical decisions based on pathology reports.
The following management framework was presented, focusing on removing high-risk lesions while avoiding unnecessary surgery for low-grade lesions:
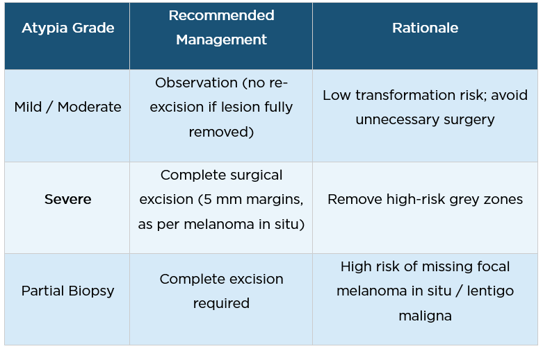
The following summarizes the clinical approach advocated during the workshop:
Reports written by Dr Joël Claveau (Dermatologist, Quebec)
Presented by Eric Lawrence Simpson, Portland, Oregon
Published in JAMA Dermatology (Okereke R et al., JAMA Dermatol. 2026 Feb;e255912), this patient decision aid was developed using standard co-design methodology, built with patients, and validated across four systemic treatment categories. It is intended to initiate shared decision-making conversations — not to serve as a definitive clinical reference.
The tool presents the four main options for moderate-to-severe AD in accessible, non-technical language:
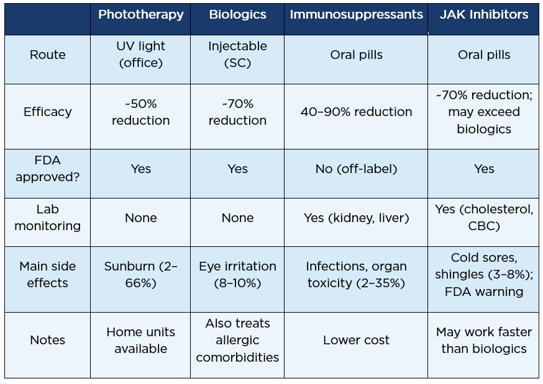
A UK study (N=80; Bajaj et al., Clin Exp Dermatol 2026) evaluated the course of atopic eczema up to 1 year following completion of narrowband UVB (NBUVB) phototherapy (2×/week). Mean age 36.4 years; moderate-to-severe AD; 46 patients completed 12 months of follow-up. Results confirmed sustained benefit post-treatment, though the study overlapped with the COVID era (affecting treatment delivery) and reported 5 adverse effects. Phototherapy remains an effective, cost-accessible option particularly suited to patients unable to access or afford newer systemic agents, with home units now commercially available.
General Class Characteristics
Biologics (dupilumab, tralokinumab, lebrikizumab, nemolizumab) are now the most common first-line systemic option for moderate-to-severe AD. They target the type 2 inflammatory pathway, require no routine laboratory monitoring, and can be used with or without topical therapy. Dosing intervals can be extended in good responders. They are not potent enough to rapidly control destabilized disease (e.g., following prednisone rebound).
Dupilumab (IL-4Rα): Specific Considerations
Pregnancy data (meta-analysis, 15 studies, 115 pregnancies; Sanchez-Garcia et al., Acta Derm Venereol 2025): 18.9% spontaneous abortion rate, no congenital abnormalities; propensity-matched cohort (293 pregnancies) showed no increase in adverse pregnancy outcomes with dupilumab (APO 12.1% vs. 22.7%; HR 0.53), with significantly reduced premature labor (HR 0.11).
Dupilumab/Tralokinumab and Facial Redness
A recognized adverse effect of both dupilumab and tralokinumab is paradoxic head and neck erythema (also termed new regional dermatosis or persistent facial dermatitis), now added to product labeling. Both agents nonetheless improve head and neck AD scores significantly (−57–68% reduction in EASI head and neck scores across SOLO and CAFÉ trial data; similar sustained response with tralokinumab in ECZTRA 1&2 at 52 weeks). When facial redness occurs or persists, the differential diagnosis includes refractory AD, contact dermatitis, steroid withdrawal/rosacea-like reaction, biologic-induced reaction, or seborrheic dermatitis/psoriasis. Management of dupilumab-induced facial redness: continue dupilumab; first-line addition is PRN topical ruxolitinib. Other options include roflumilast, tapinarof, tacrolimus, pimecrolimus, or crisaborole. Additional considerations: patch testing, oral fluconazole for Malassezia overgrowth, or topical steroid taper.
Dupilumab and Cutaneous T-Cell Lymphoma (CTCL) Risk
Several case reports have raised concern about CTCL unmasked by dupilumab or tralokinumab in patients with severe AD (itself a recognized CTCL risk factor). A TriNetX retrospective cohort study suggested a 2–4× increase in CTCL with dupilumab, but received significant methodological criticism (no adjustment for disease severity; comparison group included all AD severities; five published letters to the editor). A more robust large-scale study (Kridin et al., Front. Med. 2026; 423,306 AD patients, 18,025,971 controls) found AD carries a 0.15% risk of CTCL (HR 9.46), but dupilumab treatment was not associated with any increase in lymphoma risk (HR 0.90 for CTCL; HR 0.92 excluding Sézary disease). Dupilumab may, if anything, be protective (significantly reduced risk of Sézary disease, NHL, and multiple myeloma). Current guidance: maintain vigilance but do not withhold dupilumab solely on the basis of lymphoma concern.
Tralokinumab and Lebrikizumab (IL-13 Inhibitors): Recent Data
Tralokinumab and lebrikizumab are IL-13–selective monoclonal antibodies approved for moderate-to-severe AD in adults and adolescents (tralokinumab) or adults (lebrikizumab). Unlike dupilumab, they do not block IL-4 signalling, which may be relevant in specific clinical contexts (see Arthralgias, below). Recent data presented at AAD 2026 provide important updates on their long-term efficacy, response durability, and adverse-effect profile.
Lebrikizumab and head/neck/face dermatitis (pooled ADvocate 1&2 and ADhere): Lebrikizumab significantly improved facial dermatitis and did not increase treatment-emergent facial erythema. In pooled ADvocate 1&2 (N=409 active vs. N=229 placebo), 59.7% of lebrikizumab-treated patients reported facial dermatitis as “improved or cleared” vs. 26.6% on placebo, with 21% achieving complete clearance. In ADhere (N=105 active vs. N=39 placebo), 68.6% improved or cleared vs. 46.2%, with 31% fully cleared.
The key clinical message: facial AD responds well to lebrikizumab, but complete clearance of the face remains difficult with biologics alone and combination strategies may be needed (Murase JE et al., J Dermatolog Treat 2025;36(1):2492188).
Biologics and Inflammatory Arthritis/Arthralgias
Inflammatory arthritis and arthralgias are increasingly recognized as clinically relevant adverse effects of biologics used in AD, with important differences across drug classes.
Three JAK inhibitors – upadacitinib (UPA, JAK1-selective), abrocitinib (ABRO, JAK1-selective), and baricitinib (JAK1/2) – are approved for moderate-to-severe AD.
JAK inhibitors generally demonstrate faster onset of action, deeper endpoint attainment (e.g., EASI-90), and superior itch relief compared to biologics, at the cost of a more complex safety profile requiring monitoring and FDA boxed warnings. Head-to-head data against dupilumab are now available.
Reports written by Dr Joël Claveau (Dermatologist, Quebec)
Speakers: Harvey Lui, Tashmeeta Ahad, Benjamin F. Chong, Sally Ibbotson, Sunil Kalia
Pathogenesis
Drug-induced photosensitivity results from the photochemical interaction between ultraviolet radiation and a circulating or topically applied photosensitizing agent. Upon UV absorption, the drug undergoes photoactivation and generates reactive oxygen species through two distinct pathways:
Type 1 and type 2 photo-oxidation. Individual susceptibility is idiosyncratic and modulated by polymorphisms in drug metabolism, antioxidant defense, and transporter genes.
Spectrum of Clinical Presentations
The clinical manifestations of drug photosensitivity are diverse. Phototoxic reactions present as an exaggerated sunburn confined to UV-exposed sites, characteristically accompanied by a burning or stinging prodrome, and are dose-dependent rather than immunologically mediated. Photoallergic reactions are delayed-type hypersensitivity responses that may extend beyond the irradiated field and persist after drug cessation. Lichenoid eruptions manifest as flat-topped papules in a photo-distributed pattern. Pseudoporphyria mimics porphyria cutanea tarda with skin fragility and bullae on the dorsal hands, most commonly associated with naproxen, tetracyclines, furosemide, and sulphonylureas. Photo-onycholysis (distal nail plate separation) is characteristic of fluoroquinolones and tetracyclines. Dyspigmentation (blue-grey hyperpigmentation in photo-exposed areas) is a hallmark of amiodarone, chlorpromazine, and fluoroquinolones. Additional manifestations include drug-induced subacute lupus erythematosus (thiazides, ACE inhibitors, TNF-α antagonists, NSAIDs), photo-telangiectasia (calcium channel antagonists), and photocarcinogenesis with long-term malignancy risk elevation.
Photocarcinogenesis
Chronic photo-oxidative DNA damage induced by certain drugs confers an increased risk of non-melanoma skin cancers. Drugs with established photocarcinogenic potential include psoralens, voriconazole, azathioprine, and vemurafenib. Equivocal evidence exists for thiazide diuretics, antihypertensives, statins, and anti-epileptics, underscoring the importance of ongoing pharmacovigilance.
Photoinvestigation & Action Spectra
Monochromator phototesting determines the minimal erythema dose (MED) at discrete wavelengths and delineates the action spectrum of a patient's photosensitivity. In confirmed drug photosensitivity, abnormally reduced MED values are most frequent in the UVA range, with peak incidence around 330 nm, though UVB and visible light (VL, 400–700 nm) sensitivity may co-exist. Phototesting supports diagnosis but must be interpreted alongside the clinical history and drug initiation timeline, as results can normalize after drug withdrawal.
Common Drug Culprits
The most frequently implicated drug classes include thiazide diuretics (bendroflumethiazide, hydrochlorothiazide), tetracycline antibiotics, fluoroquinolones, NSAIDs (naproxen, ibuprofen), antifungals (voriconazole, terbinafine), proton pump inhibitors (omeprazole, lansoprazole), ACE inhibitors (lisinopril, ramipril), antimalarials (quinine, hydroxychloroquine), statins (simvastatin, atorvastatin), and amiodarone. Clinical phenotype varies by drug class: voriconazole predominantly causes phototoxicity and photocarcinogenesis; quinine and hydroxychloroquine favour eczematous and lichenoid reactions; amiodarone is uniquely associated with blue-grey dyspigmentation.
Management
First-line management involves cessation or substitution of the culprit drug, coordinated with the prescribing physician. Resolution may require 6–12 months depending on drug half-life and tissue accumulation (particularly relevant for amiodarone). Strict photoprotection is essential: broad-spectrum (UVA/UVB) sunscreens, photoprotective clothing, UV-blocking window films, and avoidance of peak solar irradiance hours. Vitamin D supplementation should be prescribed given the requirement for long-term photoavoidance. Diagnostic limitations include polypharmacy, the absence of a definitive confirmatory test, and feasibility constraints for repeat phototesting.
Diagnostic Pearls
Consider drug photosensitivity when eruption is restricted to UV-exposed anatomical sites with a burning prodrome. Establish temporal correlation with drug initiation. Action spectra are predominantly UVA. Early diagnosis is critical as this condition is potentially reversible with significant impact on quality of life (QoL).
Photodermatoses encompass a heterogeneous group of disorders triggered or exacerbated by UV radiation, with distinct epidemiological, clinical, and therapeutic nuances observed across skin phototypes. Polymorphous light eruption (PMLE) represents the most common immune-mediated photodermatosis, affecting all races and phototypes, with an onset typically within the first three decades of life. Notably, PMLE demonstrates significantly higher prevalence in Black compared to Caucasian population. Clinically, lesions are variable but tend to be monomorphic within individual patients (presenting as papules or papulovesicles) and characteristically spare the face due to photohardening. Dark-skinned individuals are more likely to develop lesions on the neck and to exhibit pinpoint papular or lichenoid variants, which may be misidentified as lichen nitidus or papular eczema. Management of PMLE includes photoprotection, topical corticosteroids, oral therapies (systemic corticosteroids, hydroxychloroquine, JAK inhibitors), and photohardening. Treatment patterns differ by skin type, with darker-skinned patients more frequently treated with combined topical corticosteroids and sunscreen and less likely to receive systemic therapy or NB-UVB photohardening.
Actinic prurigo (AP) is a photodermatosis predominantly affecting Native American and Native Alaskan populations, typically presenting in childhood, though adult-onset variants have been described in Asian populations. Its pathogenesis remains incompletely understood but is thought to involve a delayed-type hypersensitivity mechanism with a proposed genetic susceptibility linked to the HLA DRB1*0407 polymorphism. Clinically, AP manifests as a polymorphous pruritic papular, nodular, or plaque-like photodistributed eruption, with characteristic mucosal involvement including lip edema, ulceration, scaling, and hyperpigmentation (cheilitis), as well as ocular features such as photophobia, pterygium, pinguecula, and hyperemia. Management is multimodal, encompassing photoprotection, topical corticosteroids, and systemic agents including oral corticosteroids, thalidomide, and pentoxifylline. Emerging therapies such as dupilumab and JAK inhibitors have shown promise.
Chronic actinic dermatitis (CAD) typically presents in the fourth to sixth decade of life and can occur across all skin phototypes, with a predilection for darker phototypes. Sex-based differences in prevalence are notable: male predominance has been observed in Caucasian and Asian populations, whereas a female predominance is observed in Black populations. Pathogenetically, CAD is considered a delayed hypersensitivity reaction to a photoinduced neoantigen, resulting in persistent inflammation. Its action spectrum spans both UVB and UVA ranges, and it demonstrates a significant association with allergic and photoallergic contact dermatitis; sunscreen components are frequently implicated, with para-phenylenediamine sensitization being more common in skin of colour. Clinically, CAD presents as pruritic eczematous, erythematous patches to plaques on photoexposed areas, with lichenification and pigmentary alteration. In chronic cases, involvement may extend to non-photoexposed areas.
Management requires photo/patch testing to identify relevant allergens, strict photoprotection, topical agents (corticosteroids, calcineurin inhibitors), and systemic therapy (oral corticosteroids, azathioprine, mycophenolate mofetil, cyclosporine, dupilumab).
Photoaggravated disorders encompass conditions where UV exposure worsens underlying disease. Different subtypes of lupus erythematosus is a prominent example. In Black populations, DLE is disproportionately prevalent, and serologic profiles including elevated anti-dsDNA, anti-Smith, and anti-RNP antibodies are more frequently observed. In Latin American populations, lupus is associated with increased photosensitivity, livedo reticularis, and vascular thrombosis. Lichen planus variants also exhibit racial predilections: childhood lichen planus and actinic lichen planus are more common in Black, Middle Eastern, and Indian populations, while lichen planus pigmentosus predominates in Asian, Middle Eastern, and Black individuals. Chemical and drug-induced photosensitivity, particularly diltiazem-induced photoeruption, is primarily reported in SOC, manifesting as reticulated hyperpigmented macules in blue-grey to brown tones on the face, with pigmentation onset ranging from 1.5 to 150 months after drug initiation.
Photoprotection constitutes a cornerstone of management for all photodermatoses, yet SOC populations face unique barriers to its consistent application. SOC individuals tend to prefer sun-protective clothing or shade-seeking over sunscreen, and reported sunscreen use remains consistently lower in SOC compared to white populations. Application amounts frequently fall far below the recommended 2 mg/cm², often representing only one-quarter to one-half of the effective dose, and reapplication is challenging in practice. Sunscreen products specifically formulated or marketed for SOC individuals may cost up to three times more than those marketed for lighter skin, compounding access inequities.
Secondary photoprotection using oral agents such as Polypodium leucotomos extract has demonstrated benefit in PMLE by reducing clinical symptoms and UV response, with smaller case reports supporting efficacy in SCLE, actinic prurigo, solar urticaria, DLE, and erythropoietic protoporphyria. Special considerations in managing photodermatoses in SOC include the heightened risk of post-inflammatory dyspigmentation, which should be addressed secondarily.
Photosensitivity disorders constitute a diagnostically challenging group of conditions requiring systematic clinical and laboratory evaluation. The differential diagnosis of photosensitivity broadly encompasses idiopathic photodermatoses (polymorphous light eruption, solar urticaria, chronic actinic dermatitis, actinic prurigo, hydroa vacciniforme), drug-induced photosensitivity, photoaggravated connective tissue diseases, metabolic disorders (porphyrias), and genetic conditions such as xeroderma pigmentosum. A structured diagnostic approach integrating a comprehensive history, physical examination, targeted laboratory investigations, skin biopsy, genetic testing where indicated, and phototesting is essential to accurately characterize the underlying condition.
History-taking is the cornerstone of photosensitivity evaluation and should address several key dimensions: the localization of lesions (photodistributed vs. non-photodistributed); drug and environmental exposure history; the presence of a preexisting skin disease; whether sunscreens ameliorate symptoms; seasonal variation suggestive of photohardening; and associated systemic symptoms.
The temporal pattern of the reaction provides further diagnostic guidance and should be characterized according to onset (immediate vs. delayed vs. occult), evolution (transient over hours to days vs. persistent and chronic), and symptom type (rash, pruritus, pain or burning, and skin fragility). Physical examination must prioritize localization, as photodistributed involvement of the upper eyelids, periauricular folds, forehead, submental region, and dorsal hands is a critical diagnostic clue. It is important to recognize that photosensitive patients often present without visible lesions at the time of examination, often necessitating the use of photographs and phototesting.
Laboratory investigations should be directed by clinical suspicion and include autoimmune serology (ANA, ENAs, anti-dsDNA, myositis panel) and porphyrin testing via urine, stool, plasma, and red blood cells, with assessment of liver and muscle enzymes and glomerular filtration rate as clinically indicated. Urine porphyrin fluorescence under Wood's lamp examination (demonstrating pink-red fluorescence in porphyria cutanea tarda compared to normal urine) provides a rapid bedside assessment. Skin biopsy does not confirm photosensitivity per se but may reveal characteristic histopathological patterns including those of polymorphous light eruption, connective tissue disease, eczematous dermatitis, or bullous disorders with direct immunofluorescence positivity in porphyria cutanea tarda. Genetic testing is reserved for conditions such as xeroderma pigmentosum and chromosomal disorders.
Phototesting is indicated when the clinical history is equivocal, when quantification of the degree of light sensitivity is required, or when determination of the action spectrum is necessary for diagnosis or management. Three principal phototesting approaches exist: minimum erythema dose (MED) determination (applicable to chronic actinic dermatitis and drug photosensitivity), photoprovocation testing (applicable to PMLE and solar urticaria), and photopatch testing (for photocontact dermatitis, combining patch testing to a relevant allergen with UVA irradiation).
Office-based phototesting can be performed using daylight (UVB, UVA, visible spectrum), phototherapy units (UVB, UVA), or slide and data projectors for visible light. In chronic actinic dermatitis, MED determination characteristically reveals markedly reduced MEDs to both UVA and UVB. Photoprovocation of PMLE is performed using incremental UVA doses (typically 20 J/cm²) applied daily over three consecutive days, with papular or erythematous lesions constituting a positive result. Solar urticaria can be provoked using UVB, UVA, or visible light, with urticarial lesions appearing within minutes of irradiation; serial minimal urticaria doses (MUDs) quantify the threshold and action spectrum. Photopatch testing is indicated for photocontact dermatitis and involves irradiation with UVA following standard patch application.
The interpretation of phototesting results should be integrated within the broader clinical context. A summary of key phototesting patterns across conditions is as follows: PMLE typically shows no reduction in MED but a positive photoprovocation response manifesting as papules and erythema; drug photosensitivity may demonstrate a reduced UVB MED and a positive UVA photoprovocation response with erythema and edema; chronic actinic dermatitis is characterized by markedly reduced MEDs to both UVB and UVA, with photoprovocation variably positive; solar urticaria shows variable MED reduction but a consistently positive photoprovocation response to UVB, UVA, or visible light producing urticaria; and photocontact dermatitis is diagnosed by a positive photopatch test to the relevant allergen combined with UVA, producing eczematous or erythematous changes. Notably, in treatment-resistant solar urticaria, plasma exchange has been employed as a therapeutic modality, and post-treatment photoprovocation may confirm resolution of the urticarial response. These practical phototesting approaches, when systematically applied alongside clinical and laboratory data, enable accurate diagnosis and informed management of photosensitivity disorders.



Bioderma Congress Reports AAD 2025