3 professionals
Bioderma Congress Reports IMCAS 2026
Bioderma Congress Reports IMCAS 2026
Get access to exclusive dermatological services to increase your professionnal knowledge: +500 pathology visuals, clinical cases, expert videos
Benefit from valuable features: audio listening, materials to be shared with your patients
Stay informed about the upcoming events and webinars, latest scientific publications and product innovations
Already have an account? login now
Reports written by Dr. Lev NAIDOO (South Africa), Dr. Dimitris MOTSIOS (Greece) and Dr. Lidiya TODOROVA (Bulgarie).

Related topics
Report written by Dr Lidiya-Todorova (Dermatologist, Bulgaria)
Chairs: Dr. Brunilda BARDHI (Dermatologist, Albania) and Dr. Manuel Dario FRANCO (Dermatologist, Colombia)
Speakers: Dr. Brunilda BARDHI (Dermatologist, Albania), Dr. Monika Fida (Dermatologist, Albania) and Prof. Antonio A G Massa (Dermatologist, Portugal)
About the session
One of the most intriguing sessions at the event was “Clinical dermatology and aesthetic considerations”, for its focus on the evolving interface between clinical dermatology and aesthetic practice. The session underlined the way therapeutic strategies integrate medical efficacy and addressed the growing demand for holistic dermatologic management, where skin health and cosmetic impact are closely interconnected.
The session was chaired by Dr. Brunilda Bardhi (Albania) and Dr. Manuel Dario Franco (Colombia), both experienced dermatologists recognized for their contributions to clinical and aesthetic dermatology. Their combined international perspectives set the stage for a dynamic discussion on how dermatologists can balance medical management with aesthetic expectations in daily practice.
Acne scar: an aesthetic or a derm management
Speaker: Dr. Brunilda BARDHI (Dermatologist, Albania)
The first lecture, delivered by Dr. Brunilda Bardhi, engaged in the topic of acne scarring treatment as a combination of aesthetic and clinical approach. In her presentation, Dr. Bardhi addressed the dual nature of acne scarring. She highlighted that while often perceived primarily as a cosmetic concern, acne scars represent a significant dermatological condition with psychosocial impact. The presenter also emphasised the importance of early intervention, individualized treatment planning, and the integration of medical and procedural approaches for expected cosmetic results.
Acne scarring is treated in medical settings and aims at cosmetic improvement rather than medical necessity. However, acne scarring remains a condition that demands clinical assessment, morphological classification, and evidence-based intervention. It should therefore be managed by trained dermatologists capable of integrating medical knowledge with aesthetic judgment.
According to the lecturer, who is also an experienced practicing aesthetic dermatologist, the management primarily focuses on visible improvement and may include injectables, energy-based devices, skin resurfacing procedures, and combination therapies. However, treatment of acne scars is inherently complex and must be individualized.
The therapeutic approach is primarily determined by scar morphology (ice pick, boxcar, rolling), as well as scar severity, skin type, patient expectations, cost considerations, and acceptable downtime.
Discussing the range of interventions available for facial atrophic acne scars, these include:
Nevertheless, the key message of the lecture was that there is no currently available modality that can completely eliminate acne scarring. Optimal outcomes are generally achieved through combination approaches, and the establishment of realistic expectations is essential. Improvement and not total resolution is the therapeutic goal.
Dr. Bardhi concluded that dermatology provides the scientific and clinical foundation for acne scar management, while aesthetic interventions enhance outcomes.
Red Flags on the skin – Diagnostic challenges in erythematous lesions
Speaker: Dr. Monika Fida (Dermatologist, Albania)
Reporter’s subtitle: What’s new in the treatment of facial erythema ?
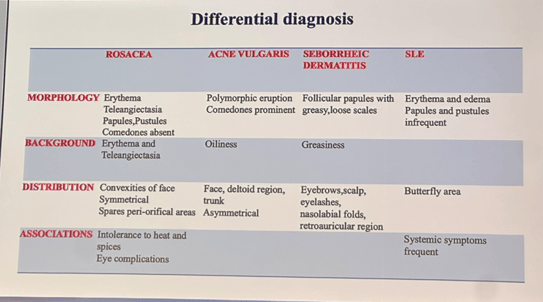
In this very detailed lecture of Dr. Fida, she discussed facial erythema, or “red face” as a visible clinical finding that does not represent a single cutaneous entity. It may result from a broad spectrum of conditions like rosacea, atopic dermatitis, demodicosis, dermatomyositis, lupus erythematosus, allergic contact dermatitis, drug-induced erythema, and carcinoid syndrome. Importantly, clinicians encounter not one uniform presentation but multiple “shades of red”, reflecting distinct pathogenic mechanisms and diagnoses [1].
Facial erythema may be classified as:
Given the broad differential diagnosis, evaluation should be guided by the onset and evolution of symptoms, characteristics of the erythema, associated functional signs, and systemic manifestations.
The lecturer started with the management of rosacea, which is phenotype-driven, as shown in the table below:

The 2019 ROSCO panel recommends alpha-adrenergic agonists (topical brimonidine, oxymetazoline) for transient or persistent erythema, noting the risk of rebound flushing. Oral beta-blockers (e.g., carvedilol) and clonidine may reduce the transient redness as well. Persistent erythema may also benefit from IPL or vascular lasers.
Seborrheic dermatitis presents with thin greasy scales on pink plaques, often involving the mid-face, scalp, and folds [3]. Management of the condition includes patient education and appropriate skincare. Acute flares may be treated with low-potency topical corticosteroids (1–2 weeks).
Topical antifungals (ketoconazole, ciclopirox) target Malassezia species; resistant cases may require zinc pyrithione or selenium sulfide. Calcineurin inhibitors are useful when repeated steroid courses are needed. In refractory adult cases, oral itraconazole, phototherapy, or low-dose isotretinoin may be considered. The new treatment options include roflumilast foam 0.3% which has demonstrated efficacy and favourable tolerability suggesting a promising nonsteroidal alternative [4].
Third in the list is the atopic dermatitis (AD). Therapeutic advances include topical and oral Janus kinase inhibitors, crisaborole and biologics. On the other hand, the management of allergic contact dermatitis (ACD) centres on allergen identification and their strict avoidance. Although topical corticosteroids are first-line for localized lesions, calcineurin inhibitors or PDE-4 inhibitors are preferred for the face, eyelids, and folds. Common in young women, perioral (periorificial) dermatitis presents with 1–2 mm erythematous papules or papulopustules around the mouth, sparing the vermilion border, often with burning and scaling [5]. In this case, “zero therapy” (discontinuation of topical steroids and cosmetics) is foundational. Mild disease responds to topical erythromycin, clindamycin, metronidazole, pimecrolimus, azelaic acid, or adapalene. Moderate–severe cases may require oral tetracyclines (sub-antimicrobial dose), clarithromycin, or erythromycin (pregnancy, children). Low-dose isotretinoin is reserved for refractory cases.
New in the treatment of psoriasis is crisaborole 2% for sensitive areas like the face, but more data is needed to confirm its efficacy [6].
Managing facial erythema remains challenging due to its broad differential diagnosis and overlapping clinical features. Success depends on precise diagnosis, individualized therapy, patient education, and long-term adherence to therapy.
Literature cited in this lecture:
Spironolactone – How I use it successfully in dermatology
Speaker: Prof. Antonio A G Massa (Dermatologist, Portugal)
Spironolactone in Dermatology: From Antihypertensive to Anti-Androgen Cornerstone
Prof. Antonio A G Massa, undoubtedly a very emotional speaker with years of experience, gave a memorable lecture on spironolactone usage in dermatology. He started with the historical perspective: spironolactone was synthesized in 1957 and received FDA approval in 1960. Initially developed during research on aldosterone-blocking agents, it became the first clinically useful mineralocorticoid receptor antagonist. Its original indication was as a potassium-sparing diuretic and antihypertensive agent, acting through antagonism of the mineralocorticoid receptor to promote sodium and water excretion while retaining potassium.
In the 1980s, dermatologists observed that female patients treated for hypertension experienced significant improvement in acne and hirsutism. This led to its widespread off-label adoption in dermatology. Its anti-androgenic properties—identified after market introduction—expanded its indications to include hormonal acne, androgenetic alopecia, and other androgen-mediated dermatoses.
But which are the approved and off-label indications of the drug? FDA currently approves spironolactone for heart failure, hypertension, oedema and primary hyperaldosteronism. All of the dermatological applications are off-label, and these include:
It is particularly useful in women with HS flares around menstruation or with coexisting acne/hirsutism. It is not a substitute for biologics in moderate-to-severe HS but serves as a valuable adjunct. It is often combined with weight control, smoking cessation, topical clindamycin, doxycycline (initial phase), hormonal contraception, and biologics (e.g., adalimumab, secukinumab) in advanced disease.
Next, Prof. Massa discussed the general principles of dosing; and gave practical advice on the topic. First, the dermatologist should start low and titrate with typically 50 mg/day (e.g., for a 60 kg woman) for the first 4–6 weeks. Then the dosage could be increased to 100 mg/day if tolerated.
Some patients require 150–200 mg/day for optimal response.
In terms of posology advise, the lecturer suggested patients to be educated to take the drug with food to improve absorption and reduce gastrointestinal discomfort. Preferably, they should take it in the morning to minimize nocturia.
Clinical improvement in acne typically requires ~3 months, reflecting hormonal cycle modulation. Spironolactone in contraindicated in pregnancy.
Sometimes patients need to combine treatments for optimal results compared to monotherapy. Spironolactone could be combined with oral contraceptives to improve menstrual regularity, enhance anti-androgenic effect and at the same time provide contraception. For the treatment of androgenetic alopecia, it could be combined with topical 5% or oral minoxidil (0.5–4 mg/day) for optimal results.
Less frequent but relevant dermatological situations, where spironolactone could be useful include:
Spironolactone represents a prime example of therapeutic repurposing: a cardiovascular drug that has become a cornerstone systemic therapy in female dermatology, particularly for androgen-mediated disorders.
Report written by Dr Lidiya-Todorova (Dermatologist, Bulgaria)
Chairs: Dr. Jean CARRUTHERS (Oculoplastic Surgeon, Canada) and Prof Hassan GALADARI (Dermatologist, United Arab Emirates)
Speakers: Dr. Marco Rocha (Dermatologist, Brazil), Dr. Todd SCHLESINGER and Dr. Diane Irvine Duncan (Plastic surgeon, USA)
Lasers and the Skin Microbiome: Toward a New Era of Microbiome-Conscious Aesthetic Dermatology
Speaker: Dr. Marco Rocha (Dermatologist, Brazil)
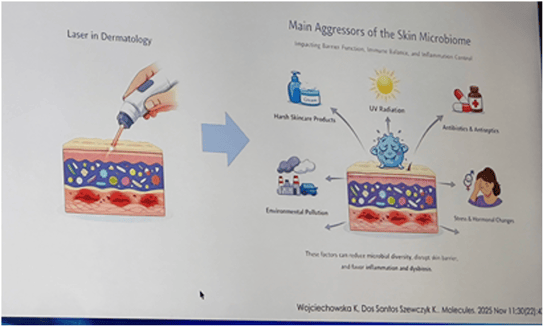
Dr. Marco Rocha addressed a timely and practice-changing question: in the era of microbiome science, are current post-CO₂ laser protocols sufficient to optimize healing? Emerging data suggests that laser procedures must be reconsidered. The reason for that: the microbiome!
Recent microbiological research highlights the regulatory role of the cutaneous ecosystem. Cutibacterium acnes functions as a key regulator of skin pH and immune homeostasis, while Staphylococcus epidermidis acts as a post-procedure “biological guardian,” supporting repair and microbial defence. In addition, the skin phageome serves as an ecological brake, preventing inflammatory overactivation and microbial imbalance. These insights are clinically relevant: a balanced microbiome promotes efficient wound healing, whereas dysbiosis disrupts the barrier and delays tissue repair [1]. Commensals such as S. epidermidis enhance healing and limit opportunistic infection, while microbial imbalance increases inflammation and delays recovery [2].
The degree of microbiome disruption varies according to laser modality:

Dr. Rocha therefore highlighted that these differences necessitate a structured, microbiome-conscious approach when a laser procedure or procedures are planned.
First, patient assessment is crucial for pre-evaluation. Prior to the laser treatment, doctor’s evaluation should include history of antibiotic use, inflammatory dermatoses (rosacea, atopic dermatitis, active acne), skin barrier integrity, and prior post-procedure complications. These factors may predispose to dysbiosis and impaired healing. Secondly, protocols should prioritize the lowest effective energy and longer intervals between sessions to reduce cumulative microbial and barrier disruption. Next, barrier repair must be the first priority. Aggressive cleansing and unnecessary antimicrobial use should be avoided to prevent further ecological imbalance. At last but not least, adjunctive use of probiotics, prebiotics, and postbiotics, along with maintenance of hydration and lipid balance, may support microbial homeostasis and recovery [3].
Emerging clinical evidence supports this approach. In post-procedure recovery, Staphylococcus epidermidis promotes wound healing and protects against opportunistic pathogens, reducing recovery time and minimizing infection risk. In pigmentation disorders, Lactobacillus fermentum has been shown to reduce oxidative stress and inhibit melanogenesis, with early data suggesting reduced hyperpigmentation when combined with topical antioxidants [3].
Athanasiou et al. demonstrated that fractional CO₂ laser combined with postbiotic-enriched moisturizers preserves microbial diversity, reduces dysbiosis, and supports barrier repair. Manolis et al. reported that probiotic-enriched serums following CO₂ resurfacing reduced infection risk and accelerated restoration of microbial balance. Together, these findings suggest that when integrated into microbiome-conscious protocols, fractional laser treatments can enhance results while maintaining microbial homeostasis [4,5].
Personalized aesthetic medicine is advancing through microbiome analysis. AI-driven platforms integrating microbiome and dermatologic data may enable adaptive protocols that optimize efficacy, predict risk, and preserve microbial balance across pre-, intra-, and post-procedure phases [3].
Dr. Rocha also suggested what every medical persona should change in their daily practice when using lasers and having microbiome-conscious protocols. First, there should be less aggressive energy stacking and smarter spacing between sessions. Then, a barrier-first post-care should be considered. Avoidance of unnecessary antimicrobials and consideration of microbiome-supportive skincare is mandatory.
The Brazilian lecturer concluded that aesthetic dermatology is entering a new era: one in which laser efficacy and microbial ecology are no longer separate considerations, but interdependent determinants of clinical success.
Literature used in the lecture:
K-Beauty – Deciphering the Global Sensation
Speaker: Dr. Todd SCHLESINGER
Some historical facts regarding K-Beauty
Dr. Todd Schlesinger discussed one of the hottest topics in dermatology currently, namely the secrets of Korean beauty cosmetics. He started his lecture with the historical foundations and the origins of K-Beauty, which date back more than 2000 years ago, with a longstanding emphasis on pure, natural skincare ingredients and a philosophy centred on hydration, balance, and barrier care. Traditional ingredients included rice water, camellia oil, and herbs such as ginseng, mugwort, and green tea.
Surprisingly for the audience, the lecturer continued that the global influence is not new but rather began in 1870 with the introduction of Western cosmetics to Korea. During the Japanese colonial era (1910–1945), Japanese brands dominated the Korean market. In 1983, increased imports allowed greater consumer access to overseas cosmetic products.
Bakgabun, launched in 1915, was Korea’s first cosmetics brand, offering a whitening powder that gained popularity due to its affordability and portability.
After liberation in 1945, Korea’s cosmetics industry expanded rapidly. In 1954, Pacific Chemical (now AmorePacific) established Korea’s first cosmetics research laboratory, recruiting leading researchers. Between the 1960s and 1980s, rapid industrialization fuelled innovation. Early forms of emulsions, toners, and nourishing creams became popular. In 1964, AmorePacific launched Korea’s first makeup campaign, “Oh My Love,” emphasizing makeup styles tailored to time, purpose, and place. Eventually, by 1971, Korea’s first beauty exports included more than 20 cosmetic products.
Modern K-Beauty growth reflects a blend of traditional values and scientific research. Its core characteristics include custom and holistic approach, layered skin care routines, and barrier-focused care. Innovative products such as BB creams, sheet masks, and cushion compacts have gained global recognition. The influence of K-Beauty market can be easily evaluated by sales rising each year with global Korean skin care market projected to reach $31.82 billion by 2033. As a matter of fact, in 2025, South Korea surpassed the United States to become the world’s second-largest cosmetics exporter after France, and in 2024 it surpassed France as the number one country of cosmetic imports in the U.S.
What makes the K-Beauty cosmetics so popular, likable and distinctive? Namely, the ingredients. K-Beauty formulations often incorporate traditional Korean herbal medicines and unique bioactive components not commonly found in Western products. Some of the most popular ingredients include:
What is the “Hallyu” effect?
“Hallyu”, as part of the Korean cultural wave, including K-pop, cinema, fashion, and cuisine, is a philosophy emphasizing hydration, even tone, and radiance. Market expansion has been amplified by social media. The number of cosmetic sellers doubled from 14,000 in 2019 to 28,000 in 2024, driven in part by platforms such as TikTok, which engage Gen Z and millennial consumers.
Despite rapid growth, concerns remain regulatory differences also exist. Some products contain UV filters and active ingredients not approved by the U.S. FDA or EMA. Many consumers obtain products through international online retailers, potentially bypassing domestic regulatory oversight.
K-Beauty represents a unique fusion of tradition, innovation and cultural influence. Its global success is driven by the unique philosophy behind it and strong cultural exportation. However, continued emphasis on evidence-based validation and regulatory transparency remains essential as the market continues to expand.
Regenerative battle: biologicals vs EBDS
Speaker: Dr. Diane Irvine Duncan (Plastic surgeon, USA)
Dr. Diane Irvine Duncan, US-based board-certified plastic surgeon known for research and international education. She presented a thought-provoking comparison between two dominant forces in aesthetic medicine: energy-based devices (EBDs) and regenerative biological therapies. Rather than framing the discussion as a true opposition, she emphasized their differing mechanisms, advantages, and strategic roles in clinical practice.
EBDs remain a cornerstone of modern aesthetic medicine. They are ubiquitous in practices worldwide as they offer technology-driven, visible, and reproducible results.
A single device platform may address multiple indications. An example for such include skin tightening, resurfacing, pigment correction, and vascular lesions. Key strengths of EBD feature: immediate or short-term visible improvement, scalability across large treatment areas, clearly defined parameters and protocols. All of these characteristics have secured EBDs a dominant role in procedural aesthetics.
On the other side are the biological therapies—including platelet-based treatments, biostimulatory injectables, fat transfer, stromal vascular fraction (SVF), exosomes, growth factors, and allografting. All these therapies have evolved in parallel with EBDs since the late 1980s and 1990s. Unlike devices, biologicals do not rely on thermal or mechanical injury. Their mechanism is based on cell signalling, regenerative pathways, angiogenesis, and matrix remodelling. Fat, for instance, is increasingly understood not merely as a volumizing agent, but as metabolically active signalling tissue. Biologicals represent a shift from ablation to regeneration.
Dr. Duncan then compared the strategic advantages of EBD to biological therapies. EBD are beneficial because of the predictable tissue heating and collagen remodelling, efficient large-area treatments, established safety profiles when properly used. On the contrary, biologicals have minimal downtime, lower discomfort compared to many EBD procedures, no thermal injury and therefore reduced risk of burns or post-inflammatory hyperpigmentation (PIH), gradual, natural-looking improvements, particularly well-suited for fragile skin, higher Fitzpatrick phototypes, prevention, and maintenance strategies.
Of course, both treatments have limitations and complications. EBDs carry risks including burns, PIH and prolonged erythema or grid-pattern marks. They are also operator-dependent, and the results are variable based on the operator experience. The operator should also always have in mind patient expectations.
On the other hand, biological therapies present different challenges including variable graft survival, nodules or contour irregularities, poor technique-related complications and risk of contaminated or usage of non-standardized products.
Both approaches are sensitive in terms of technique and of course require appropriate patient selection. Another important factor in patients’ selection is the economical factor. Biological therapies typically require lower capital investment, making them accessible to smaller or emerging practices. They do not involve expensive hardware or devices. In contrast, EBDs require significant capital investment, consumables, maintenance, and upgrades. They are also associated with treatment discomfort, temporary oedema, erythema, crusting, or peeling.
By the end of her lecture, Dr. Duncan emphasized a critical principle: the clinical outcome is determined not by the intervention itself, but by the tissue response it induces. EBDs act through controlled energy delivery, inducing sub-ablative heating and collagen remodelling. Energy serves as a biological trigger. Biologicals use cells, extracellular matrix, and signalling molecules to promote angiogenesis and regeneration. Here, tissue is treated as a living system rather than a passive target.
To sum up, the “battle” between EBDs and biologicals is less about competition and more about understanding complementary mechanisms. EBDs provide structured, technology-driven tissue regeneration, while biologicals enhance regenerative signalling and tissue quality. The future of aesthetic medicine may not lie in choosing one over the other, but in strategically integrating both.
Report written by Dr Lidiya-Todorova (Dermatologist, Bulgaria)
Chairs: Dr. Nicolas KLUGER (Dermatologist, Finland) and Dr. Ximena WORTSMAN CANOVAS (Radiologist, Chile)
Speakers: Dr. Nicolas KLUGER (Dermatologist, Finland), Dr. Claudio MARASCA (Dermatologist, Italy) and Dr. Ofir ARTZI (Dermatologist, Israel)
How to manage an hidradenitis suppurativa (HS) patient in private consultation?
Speaker: Dr. Nicolas KLUGER (Dermatologist, Finland)
Hidradenitis suppurativa (HS) is a chronic, non-infectious inflammatory disease of the hair follicle characterized by recurrent painful nodules, abscesses, draining tunnels (sinus tracts), and scarring in intertriginous areas. It typically begins after puberty, is more common in women, and is strongly associated with smoking, obesity, and metabolic syndrome. HS significantly impairs quality of life, causing pain and psychosocial distress. Global prevalence is estimated between 0.67% and 1.46%
Importantly, disease severity may differ between care settings. A community-based cohort study reported that progression to severe HS was 10.4 times lower and remission rates 3.8 times higher than hospital-based estimates, highlighting differences between referral populations and real-world practice.
In this debilitating disease, Prof. Kluger highlighted the need for a structuring the HS consultation. He advised on structured consultation to avoid dispersion, and with the help of ready-to-fill standardized forms the consultation would ensure comprehensive assessment and focus on the patient’s primary concern.
An example for a structured consultation with key elements of patient’s history is as follows:
Clinical evaluation should include careful inspection and palpation. Pinching the skin may help reveal subclinical infiltration. Clinical photography is recommended for follow-up and objective assessment. Dermatologists should be aware of the international Hidradenitis Suppurativa Severity Score System (IHS-4), which evaluates inflammatory lesions only:
Severity categories:
Non-inflammatory lesions (e.g., inactive tunnels) should not be included in scoring at the time of examination. Hurley classification and modified Hurley scoring remain complementary tools in staging disease severity.
Management should be individualized based on severity, phenotype, comorbidities, and patient expectations. In acute flares short-term antibiotics may be used, including amoxicillin–clavulanic acid, second-generation cephalosporins, metronidazole, clindamycin. Emergency incision and drainage may be required in painful abscesses.
Long-term medical treatment requires antibiotics like doxycycline, tetracycline, or lymecycline; trimethoprim–sulfamethoxazole; clindamycin ± rifampicin. The retentional forms should be managed with acitretin, and if its associated with acne, isotretinoin is preferable. Adjunctive therapies include oral zinc, dapsone, intralesional corticosteroids. In case there is hormonal and metabolic concomitant diseases like PCOS/hyperandrogenism, spironolactone 100–150 mg is to be given; metabolic syndrome: metformin (e.g., 1500 mg), type 2 diabetes: semaglutide (2–2.4 mg/week). At last, lifestyle interventions such as weight control, smoking cessation are essential components of management.
Procedural and surgical options are also considered, including laser hair removal, deroofing procedures, localized or extensive surgery depending on severity. At last but not least, biologic and advanced therapies should be considered. For moderate-to-severe disease there are TNF-α inhibitors (adalimumab (ADA), infliximab (IFX); IL-17 antagonists (secukinumab (SEK), bimekizumab (BKZ); JAK inhibitors. These agents are supported by contemporary European guidelines.
HS management in private practice requires a structured, comprehensive, and patient-centred approach. Accurate severity assessment using tools such as IHS-4, systematic evaluation of comorbidities and triggers, and tailored therapeutic strategies—including medical, surgical, hormonal, metabolic, and biologic options—are essential. Understanding that community-based HS populations may differ significantly from tertiary referral cohorts further underscores the importance of individualized care and realistic therapeutic goals.
PDT, Lasers & Botulinum Toxin for the management of Hidradenitis Suppurativa: emerging treatments to be considered alongside biologic drug and surgery
Speaker: Dr. Claudio MARASCA (Dermatologist, Italy)
Unconventional approaches for Hidradenitis Suppurativa: from laser to botulinum toxin
Dr. Claudio Marasca presented emerging procedural approaches for hidradenitis suppurativa (HS), highlighting photodynamic therapy (PDT), laser-based treatments, and botulinum toxin (BTX) as adjunctive strategies to biologic therapy and surgery.
He presented a case series, which evaluated ablative fractional CO₂ laser (CO₂-AFL), alone or combined with intense pulsed light (IPL), followed by topical and intralesional triamcinolone:
All patients reported high satisfaction, and no adverse effects were observed.
These preliminary findings suggest that CO₂-AFL, particularly when combined with IPL and corticosteroid therapy, may improve both scar quality and quality of life in HS. However, larger prospective studies with standardized protocols and longer follow-up are required to validate these results.
In addition, a prospective cohort study compared fractional CO₂ laser with fractional radiofrequency microneedling for HS-associated atrophic scars, supporting the role of energy-based devices in post-inflammatory scar management.
Dr. Marasca highlighted the importance of secondary prevention. Measures such as reducing mechanical irritation, avoiding shaving, optimizing skin care, performing laser hair removal, and using fusidic acid/betamethasone cream resulted in clinical improvement in 62.5% of patients.
Botulinum toxin type A has been proposed as a treatment for HS, particularly in patients with concomitant hyperhidrosis, which may exacerbate disease activity and impair quality of life. How does BTX help in HS:
Although the exact mechanism remains speculative, the anhidrotic effect in intertriginous areas may alter bacterial growth and microbiome dynamics, potentially influencing disease pathogenesis.
A review including 16 studies and 252 HS patients found that BTX treatment improved disease severity, quality of life, and hyperhidrosis, with good tolerability. Injection protocols commonly divide the affected area into 1.5 cm² squares, administering 4 units of Botox per square.
Photodynamic therapy may benefit HS by disrupting bacterial biofilms, particularly those formed by Staphylococcus epidermidis and Staphylococcus aureus. Therefore, PDT may reduce chronic inflammation and lesion persistence.
Energy-based therapies, botulinum toxin, and PDT represent promising adjunctive strategies in HS management. While biologics and surgery remain central in moderate-to-severe disease, these emerging approaches may contribute to secondary prevention, scar improvement, symptom control and quality-of-life. Optimal protocols, and position within HS treatment algorithms should be developed to position those treatment methods in the HS management.
From Acne to Hidradenitis: Energy-Based Devices Transforming Follicular Disease Management
Speaker: Dr. Ofir ARTZI (Dermatologist, Israel)
Over the past century, dermatology has evolved through major surgical, pharmacologic, technological, and molecular innovations that have fundamentally transformed patient care. For example, the development of MOHS micrographic surgery in the 1930s represented a major breakthrough in cutaneous oncology, providing highly effective, tissue-sparing technique for the management of non-melanoma skin cancer and selected melanomas. In the mid-twentieth century, the rise of dermatopathology revolutionized diagnostic precision. Detailed histopathologic examination of skin biopsy specimens allowed for more accurate disease classification and increasingly targeted therapeutic strategies.1950s marked the introduction of topical corticosteroids, transforming the management of inflammatory dermatoses such as eczema and psoriasis. Systemic corticosteroids further expanded therapeutic options for severe inflammatory and autoimmune skin diseases.
Subsequent pharmacologic advances included: isotretinoin (1982), which improved outcomes in severe acne by targeting sebaceous gland activity and follicular keratinization. Biologic therapies (2000s), which introduced targeted immune modulation for conditions such as psoriasis and atopic dermatitis, significantly improving efficacy and safety profiles compared to traditional systemic agents. Phototherapy (1960s), including UV-based treatments, became a mainstay for diseases such as psoriasis and vitiligo, offering an effective alternative to systemic therapy.
But when was the rise of procedural and cosmetic dermatology?
From the 1980s onward, laser technology and cosmetic dermatology expanded rapidly. Laser systems enabled selective photothermolysis for vascular lesions, pigmentary disorders, and hair removal.
Non-surgical aesthetic procedures, including fillers, botulinum toxin, and chemical peels, transformed aesthetic dermatology by offering minimally invasive rejuvenation options.
Common device-based treatments now include:
Since the 1990s, teledermatology has expanded access to care, particularly in remote or underserved regions, enabling remote consultations and triage. In the 2010s, artificial intelligence (AI) began supporting dermatologic practice. AI-based image analysis tools assist in lesion recognition, skin cancer screening, and workflow optimization, enhancing diagnostic accuracy and efficiency. The advent of gene therapy and CRISPR-based technologies has opened new therapeutic possibilities for inherited skin diseases by targeting and correcting underlying genetic mutations.
Energy-based technologies continue to evolve. In acne management, radiofrequency (RF) microneedling using micro-insulated needles targets sebaceous glands, reduces pore size, and modulates inflammation. RF has demonstrated anti-inflammatory effects, potentially mediated through modulation of the NF-κB signalling pathway. It is one of the promising procedural options for HS management with over 50% success rate. From histologic precision and corticosteroid therapy to biologics, lasers, AI, and gene editing, dermatology has undergone profound transformation.
The specialty continues to integrate molecular science, device innovation, and digital technology, moving toward increasingly personalized, targeted, and minimally invasive patient care.

Report written by Dr Lidiya-Todorova (Dermatologist, Bulgaria)
Chairs: Dr. Leonor Alda GIRAO (Dermatologist, Portugal) and Dr. Nicolas KLUGER (Dermatologist, Finland),
Speakers: Dr. Nicolas KLUGER (Dermatologist, Finland)
Tattoos and Medical Myths: Evidence-Based Clarifications
Speaker: Dr. Nicolas KLUGER (Dermatologist, Finland)
This presentation of Prof. Kluger addresses persistent myths surrounding tattoos, focusing on cancer risk, medical procedures, allergic reactions, and misleading commercial claims. The objective was to distinguish evidence-based data from misinformation frequently encountered in clinical practice.
Concerns that “tattoos will give you cancer” remain widespread. However, current evidence does not support a causal association. Since 1938, approximately 45 melanoma cases reported on tattoos have been described in the literature, with a mean Breslow thickness of 2.7 mm. Notably:
A national registry study (1991–2023) including 94 melanomas in tattooed individuals found a median Breslow thickness of 0.9 mm, further arguing against systematic diagnostic delay. Several methodological biases complicate interpretation of isolated case reports, including:
Furthermore, animal studies have explored potential carcinogenic components such as benzo(a)pyrene (BaP) in black ink and 2-anisidine in red ink. While tumour acceleration has been reported in murine models, tumours often arose outside the tattooed area, and translation to human risk remains unproven.
In addition, when it comes to internal malignancies, large-scale studies show that there is no significant association between tattoos and lymphoma; no confirmed association with bladder cancer; and no convincing link with other internal cancers.
Some hypotheses even suggest possible protective behaviours (e.g., increased skin awareness), though this remains speculative. Therefore, the conclusion is that the current human data do not demonstrate a causal association between tattoos and cancer.
What are the ongoing tattoo removal myths? The claim that “a cream can remove my tattoo” is widely marketed online. These products are frequently scams. Some contain depigmenting agents such as: kojic acid, alpha-arbutin (hydroquinone derivative), dimethylmethoxy chromanyl palmitate. However, there is no scientific evidence that topical creams can remove tattoo pigment deposited in the dermis. Similarly, non-laser “new methods better than laser and surgery” lack robust clinical evidence and may carry significant risks.
Laser therapy remains the gold standard for tattoo removal.
Prof. Kluger continued and highlighted several common misconceptions. First, the epidural anaesthesia in lower back tattoos is not an absolute contraindication. Evidence does not support routine refusal. Secondly, vaccination through tattooed skin is feasible. No evidence supports avoiding injections solely due to tattoo presence. Procedures including venipuncture, peripheral and central venous catheterization, haemodialysis access, local/regional infiltration and spinal tap can generally be performed through tattooed skin when clinically indicated.
A rare case of joint infection occurred when an MRI arthrogram was performed through a 5-day-old fresh tattoo, highlighting that recently tattooed skin should be avoided due to infection risk.
How about tattoos during breastfeeding? It has been suggested that mothers should wait to get a tattoo until 9–12 months postpartum, when the infant is no longer exclusively breastfed. The concern relates to infection risk and systemic exposure rather than ink transfer through breast milk.
The belief that nickel allergy contraindicates tattooing is not supported by evidence. Standard “ink tests” or informal patch-like tests performed by tattoo artists are not reliable predictors of allergic reactions. When it comes to complications however, we should have in mind the following:
The major clinical message is that most tattoo-related fears, particularly regarding cancer, are not supported by high-quality evidence. Physicians should provide balanced, data-driven counselling and correct misinformation while acknowledging rare but documented complications.
The importance of this presentation is that it highlights the scientific rigor in evaluating tattoo safety and combating misinformation in both clinical practice and public discourse. Every dermatologist and medical professional should have in mind the following:
Report written by Dr Lidiya-Todorova (Dermatologist, Bulgaria)
Chairs: Dr. Suleima Arruda and Prof. Neil Sadick
Speakers: Ronda Farah and Pr. Neil S. Sadick
Hair Dyes: Mechanisms, Safety, and Clinical Considerations
Speaker: Ronda Farah
Hair dyeing involves either deposition of pigment onto the hair shaft or chemical alteration of intrinsic melanin. Appropriate product selection depends on patient goals, particularly desired colour and duration of effect.
When counselling patients, two key questions should be addressed:
Colour change may involve either lightening or deposit-only dyeing, which differ significantly in mechanism and degree of hair damage. Lightening typically requires bleaching followed by dyeing. Bleaching uses alkaline oxidizing agents to open the cuticle and oxidize melanin within the cortex. This process ruptures melanin granules, reduces tensile strength of hair fibres, damages the cuticle to allow dye penetration
Hydrogen peroxide is the principal oxidizing agent; higher concentrations produce greater lightening. However, peroxide alone cannot sufficiently lighten very dark hair to blonde shades, necessitating additional bleaching boosters. Because melanin destruction is required, bleaching is inherently damaging.
Which are the complications of bleaching? These include chemical burns, cuticular damage, trichoptilosis (split ends), hair fragility and hair loss.
Management includes discontinuation of chemical processing, intensive conditioning treatments, and protective measures such as petroleum jelly for chemical burns.
Dyeing without bleaching is a one-step process. When bleaching is not performed, dyeing causes comparatively less structural damage. The mechanical integrity of the hair shaft is largely preserved.
When it comes to temporary dyes, they are typically anionic and remain on the hair surface (do not penetrate the cortex). They are easily washed out and are useful for neutralizing yellow tones in grey hair or enhancing platinum hues. In addition, they do not significantly alter dark hair colour
Semi-permanent dyes on the other hand are usually cationic, attracted to negatively charged hair. Smaller molecular size allows partial penetration past the cuticle. They last approximately 3–10 washes and are used to enhance tone, add highlights, or blend grey hair. Some formulations combine semi-permanent dyes with oxidation dye precursors and alkalizing agents, extending durability to 10–20 washes.
Permanent dyes are the longest lasting and most versatile and they cannot be removed by shampooing. Those dyes use alkalizing agents to open the cuticle. They contain oxidation dye precursors, oxidizing agents, and coupling bases.p-Phenylenediamine (PPD) is a commonly used oxidation dye precursor and a well-recognized contact allergen. Selective dye application creates natural variation:
Additives in modern hair dye formulations have a protective function and may include:
Patients often ask about colour-preserving shampoos. The doctor should advise that they should avoid harsh sulphates that strip pigment and be pH-balanced to maintain cuticle closure. They also have to contain protective ingredients (e.g., antioxidants, UV filters, plant extracts) to reduce environmental fading.
For patients with PPD allergy, alternative colouring agents include bismuth citrate (black/brown tones), cassia auriculata (golden tint), indigo leaf powder (auburn tones), phyllanthus emblica (darkening effect) henna (red tones). However, natural products are not inherently allergen-free.
Beyond allergic contact dermatitis, systemic PPD poisoning, often reported in intentional ingestion, can cause: cervicofacial angioedema, tongue swelling, rhabdomyolysis, impending acute kidney injury. A mortality rate of approximately 12% has been reported in severe cases, underscoring the potential systemic toxicity of PPD. At home hair dyeing have mild tinting effects and these include:
These methods produce modest and often unpredictable results.
To conclude, the lecturer emphasised that understanding the chemical mechanisms and safety profiles of hair dyes allows clinicians to provide informed counselling on efficacy, risks, and appropriate product selection.
Trichoceuticals: Emerging Adjuncts in Hair Loss Management
Speaker: Pr. Neil S. Sadick
Trichoceuticals have been introduced as a concept at IMCAS. Trichoceuticals are hair care products containing biologically active ingredients designed to support hair and scalp health. They are not FDA-approved drugs and do not fit strictly into the categories of pharmaceuticals or cosmetics. Available in oral and topical formulations, they represent a growing sector within integrative hair restoration.
Prof. Sadick introduced the concept of Trichohealth, which emphasizes early intervention, maintenance, and restoration rather than treatment limited to visible hair loss. This approach supports a holistic, multimodal strategy integrating medical therapies, procedural interventions, lifestyle modification, and targeted trichoceuticals. Energy-based scalp and hair devices form an important complementary component.
Integral part of trichohealth are the trichoceuticals, which commonly contain:
Their proposed biological actions include anti-inflammatory effects, hormonal modulation, antioxidant activity, stem cell stimulation, pro-angiogenic effects. These mechanisms aim to influence follicular cycling, reduce perifollicular inflammation, and optimize the scalp microenvironment.
Key ingredients of the trichoceuticals include:
Next-Generation trichoceuticals therefore are those emerging formulations which include both topical cosmeceuticals and oral nutraceuticals. Topicals usually contain combinations such as saw palmetto, caffeine, resveratrol, peptides, curcumin, ashwagandha, plant stem cells, and grape seed extract.
On the other hand, oral nutraceuticals include marine collagen or marine protein extracts, curcumin, saw palmetto, ashwagandha, vitamin C, and melatonin.
Clinical data for certain marine-based supplements demonstrate improvements in hair thickness, hair count, and scalp coverage after six months of use in women with hair thinning. A randomized, controlled study in 50 adults evaluating a proprietary trichoceutical formulation assessed hair quality, scalp health, safety, and tolerability, contributing controlled evidence to this evolving field. Nevertheless, only a limited number of products are supported by robust clinical trials.
It is important to remember that trichoceuticals are not the management of hair loss, but rather a complementary cosmetics that could prevent hair loss or could add to the medical treatment of the hair condition, Therefore to be effective, trichoceuticals require identification of the underlying cause of hair loss, individualized matching of active agents to patient needs, ongoing monitoring and adjustment of treatment plans.
Trichoceuticals are perfect in combined treatments, and they can be used alongside with:
Fractional lasers may enhance follicular stem cell activation and growth factor signalling, promote follicular neogenesis, and increase transdermal penetration of topical agents while allowing precise follicular targeting with minimal collateral tissue damage. Combination strategies may improve the bioavailability and efficacy of trichoceuticals within comprehensive hair restoration programs.
Trichoceuticals represent a promising but still evolving domain requiring continued scientific validation and careful patient selection. They aim to modulate follicular cycling, inflammation, and scalp biology and may serve as useful adjuncts within multimodal hair loss management. Their combination with energy-based technologies may enhance therapeutic penetration and outcomes. Despite growing popularity, relatively few formulations are supported by high-quality clinical evidence. Clinicians should exercise caution and counsel patients critically regarding over-the-counter “hair supplements.”
Report written by Dr Dimitris Motsios (Dermatologist-Venereologist, Greece)
Speakers: Dr Shadi Kourosh (USA), Dr David Gunn (Netherlands), Dr Yuki Ogura (Japan), Dr Mukta Sachdev (India), Virginie Couturaud (France), Rishabh Kala (India) and Dr Elodie Valin (France)
Skin Longevity: Separating the Science from Snake Oil
Speaker: Dr Shadi Kourosh (USA)
Dr Kourosh opened the session by addressing the rapid proliferation of the term “skin longevity,” emphasizing the necessity of anchoring this concept in measurable biological mechanisms rather than marketing language. She proposed that true longevity in dermatology must be defined by preservation of cellular competence, structural integrity of the extracellular matrix (ECM), and sustained inflammatory equilibrium over time. A central focus of her lecture was inflammaging a chronic, low-grade inflammatory state characterized by persistent activation of NF-κB signalling and increased expression of IL-6, TNF-α, and matrix metalloproteinases (MMP-1, MMP-3). These mediators drive progressive collagen I and III degradation and elastin fragmentation, weakening dermal tensile strength [1]. Importantly, she highlighted the role of senescent fibroblasts expressing p16INK4a and components of the senescence-associated secretory phenotype (SASP), which perpetuate inflammatory amplification and ECM breakdown. Mitochondrial decline was presented as an upstream event in visible skin aging. Reduced NAD+ availability compromises SIRT1 activity, impairing genomic repair mechanisms and increasing oxidative stress burden [2]. This metabolic insufficiency accelerates collagen crosslinking through glycation and enhances dermal stiffness. The result is not simply wrinkle formation but a progressive reduction in tissue resilience. Clinically, Dr Kourosh advocated a shift from corrective strategies toward biologically preventive interventions. Barrier stabilization reduces inflammatory signalling cascades; mitochondrial support and redox balance help preserve fibroblast functionality; and early ECM protection may delay structural collapse. Rather than focusing exclusively on wrinkle depth or volume loss, dermatologists should evaluate durability, structural coherence, and inflammatory control as longevity endpoints. The overarching message was clear: skin longevity must be evidence-driven, mechanistically justified, and rooted in intrinsic biological preservation rather than accumulation of external materials.
Youthful Looks and Health
Speaker: Dr David Gunn (Netherlands)
Dr Gunn presented longitudinal epidemiological data supporting the concept that facial appearance reflects systemic biological aging. Drawing primarily from the Rotterdam Study, he demonstrated that individuals who appeared younger than their chronological age exhibited significantly lower rates of osteoporosis, chronic obstructive pulmonary disease, cataracts, and cognitive decline [3].
These associations persisted after adjustment for lifestyle factors, suggesting that perceived age reflects underlying biological processes rather than cosmetic variation alone. Further evidence from a Danish twin cohort reinforced this association. In that study, each additional perceived age year increased mortality risk by 14%, exceeding the predictive value of chronological age (11%) [4]. Importantly, controlled image manipulation experiments confirmed that intrinsic facial morphology skin texture, wrinkle distribution, dermal laxity rather than hairstyle or clothing, drove mortality prediction. Mechanistically, dermal aging mirrors systemic aging pathways. Collagen density reduction, elastin fragmentation, and glycation-induced crosslinking increase tissue stiffness and impair biomechanical recovery. Advanced glycation end-products (AGEs) accumulate with age and oxidative stress, promoting fibroblast dysfunction and vascular compromise. Mitochondrial inefficiency and chronic low-grade inflammation further contribute to microvascular fragility and ECM degradation. Dr Gunn also discussed the emerging role of AI-based morphometric analysis. High-resolution phenotyping can quantify subtle structural changes in dermal architecture, potentially transforming perceived age from a subjective metric into an objective biological biomarker. The clinical implication is significant: improving skin structure may reflect systemic health optimization. Dermatology therefore occupies a unique position at the intersection of aesthetics and preventive medicine, where visible skin changes provide early insight into organismal aging trajectories.
Next-Generation Anti-Aging Strategy for Wrinkle Improvement and Prevention
Speaker: Dr Yuki Ogura (Japan)
Dr Ogura presented a biomechanical model of wrinkle formation based on 3D elasticity mapping.
Two structural alterations were identified in aged skin: the development of an “elastic gap” between a stiffened stratum corneum and a softened papillary dermis, and selective loss of compliance within the superficial dermal layer. Mechanical simulations demonstrated that even modest increases in stratum corneum stiffness amplify stress transmission to deeper dermal compartments. This stress redistribution contributes to wrinkle propagation independent of absolute collagen quantity. A critical molecular finding involved the decline of type V collagen after age 40. Type V collagen regulates fibrillar assembly of type I collagen and contributes to dermal elasticity. Reduction in its expression alters fibril architecture and compromises biomechanical flexibility. Ex vivo studies demonstrated that retinol restored type V collagen expression within 48 hours while simultaneously reducing stratum corneum stiffness [5].Beyond its well-known effect on keratinocyte turnover, retinol appears to modulate papillary dermal biology. By influencing collagen synthesis, reducing MMP expression, and supporting dermal matrix organization, retinoids function as structural regulators rather than superficial exfoliants. Under current European regulatory concentration limits, stabilization technologies become essential to maintain efficacy at lower doses. This highlights the importance of formulation science in achieving biological impact. The lecture reframed wrinkle prevention as correction of mechanical imbalance and restoration of dermal coherence, aligning anti-aging interventions with tissue biomechanics rather than purely cosmetic smoothing.
The Longevity Aesthetic Blueprint
Speaker: Dr Mukta Sachdev (India)
Dr Sachdev described a clear shift in aesthetic dermatology from volumetric replacement toward regenerative sequencing. Patients increasingly request natural outcomes that preserve facial identity while improving tissue quality.
Regenerative approaches aim to stimulate intrinsic repair pathways rather than introduce inert bulk. Key biological mechanisms include fibroblast activation, TGF-β modulation, neoangiogenesis, and controlled inflammatory signalling. Platelet-rich plasma (PRP) delivers growth factors such as PDGF and VEGF, enhancing collagen synthesis and vascular support. Injectable platelet-rich fibrin (iPRF) provides sustained release of cytokines and growth factors. Polynucleotides (PDRN) stimulate DNA synthesis and promote angiogenesis through adenosine A2A receptor activation, supporting dermal regeneration. Biostimulatory injectables induce controlled collagenesis through mild inflammatory signalling, leading to gradual matrix remodeling. The emphasis is on tissue behaviour rather than volume accumulation. Excessive filler deposition may alter biomechanics and compromise long-term structural coherence, whereas regenerative sequencing supports progressive dermal strengthening. Dr Sachdev highlighted that early intervention sometimes termed “prejuvenation” may delay structural collapse by maintaining ECM quality before significant volume loss occurs. The overarching principle is biological coherence: interventions should respect tissue physiology, minimize inflammatory burden, and promote durable matrix organization. This approach reflects a maturation of aesthetic dermatology toward long-term structural optimization.
Reverse Aging Beyond Anti-Aging
Speaker: Virginie Couturaud (France)
Virginie Couturaud presented transcriptomic and proteomic insights into aging skin derived from single-cell analysis. Over 800 genes in keratinocytes and more than 1,000 in dermal fibroblasts demonstrated age-dependent modulation. Affected pathways included oxidative stress regulation, circadian rhythm control, mitochondrial metabolism, and ECM structural proteins. Proteostasis decline emerged as a key contributor to aging. Impaired autophagy and reduced proteasome efficiency allow accumulation of damaged proteins, accelerating cellular dysfunction. Concurrent dysregulation of NAD+ metabolism and sirtuin signalling compromises genomic stability and redox homeostasis. Cutaneous aging was also discussed in relation to systemic aging. Recent collaborative research published in Nature Aging suggested that senescent cell accumulation in skin may contribute to broader inflammatory signalling, positioning skin as an active participant in organismal aging rather than a passive reflector. The lecture emphasized precision strategies targeting redox balance, mitochondrial efficiency, and structural protein preservation. Interventions that stabilize cellular metabolism and maintain proteome integrity may extend skin healthspan. This systems-biology perspective reinforces a shift from superficial anti-aging claims toward mechanistically validated longevity interventions grounded in cellular preservation.
Genomics, Metabolomics and Personalized Skin Aging
Speaker: Rishabh Kala (India)
Rishabh Kala explored the integration of genomics and metabolomics in personalized dermatology. Genetic polymorphisms affecting inflammatory mediators, MMP expression, FLG barrier proteins, and aquaporins influence hydration, inflammatory susceptibility, and collagen turnover. Variants in BCO1 may impair conversion of beta-carotene into active vitamin A, altering retinoid responsiveness. Polymorphisms affecting vitamin C transport and recycling influence collagen hydroxylation efficiency. These predispositions partially explain inter-individual variability in treatment response. Metabolomics provides real-time insight into oxidative stress markers, glutathione availability, glycation products, and one-carbon cycle efficiency. Assessment of transsulfuration pathways and methylation capacity may reveal redox vulnerability or accelerated matrix degradation.
The combination of genetic predisposition and metabolic state enables individualized strategies targeting barrier repair, antioxidant reinforcement, and collagen preservation. Rather than applying uniform protocols, precision dermatology seeks to align interventions with biological variability, improving predictability and long-term outcomes.
Skin Microbiome and Aging
Speaker: Dr Elodie Valin (France)
Dr Valin presented large-scale microbiome data demonstrating reproducible age-related compositional shifts. Aging skin shows reduced abundance of Cutibacterium and Lactobacillus species and increased representation of anaerobic taxa. Cutibacterium metabolizes sebum lipids into free fatty acids and propionic acids, contributing to PPARα activation and maintenance of barrier integrity. Age-related decline reduces beneficial lipid metabolism and may impair hydration and inflammatory regulation. Microbial diversity shifts influence local pH, lipid composition, and immune signalling. Dysbiosis may amplify low-grade inflammation and barrier fragility, accelerating structural decline. Machine learning models applied to microbiome datasets demonstrated accurate prediction of chronological age based solely on microbial composition, reinforcing the microbiome as a biomarker of aging trajectory. Preservation of microbial balance, gentle cleansing strategies, and barrier-supportive formulations emerge as central components of longevity-oriented dermatology.
References
Summary
What struck me at IMCAS 2026 is that the entire discussion, whether it came from cosmeceuticals, injectables, devices, microbiome, or genomics, kept converging on the same destination: skin aging is being reframed as a problem of biological durability, and the future is bioregeneration and skin healthspan, not more product and not more volume. The sessions repeatedly moved away from the old correction model and toward a model where we preserve and restore what the skin is supposed to do: maintain barrier competence, regulate inflammation, sustain mitochondrial energy balance, and keep the extracellular matrix mechanically coherent over time. That is the foundation of natural results today, not minimalism as a lifestyle choice, but a clinical philosophy where the most natural outcome is the visible expression of restored physiology.
At IMCAS, this shift was explicit in the skin longevity and healthspan framing: dermatology is moving from antiaging toward healthspan, emphasizing structure, function, resilience, and prevention rather than purely aesthetic endpoints. That theme appeared again and again in different scientific languages. In the next generation antiaging strategy lecture, wrinkles were presented not as superficial lines but as visible endpoints of deeper mechanical and biological changes.
The most important message was that modern antiaging does not erase, it normalises mechanics and biology early, before the system collapses into correction. The retinol elasticity lecture then gave the most concrete mechanistic explanation of what this means: wrinkle formation was tied to a previously underrecognized mechanical imbalance, including an elastic gap between a stiffened stratum corneum and a softened dermis, and a selective softened layer in the upper dermis, both of which actively drive wrinkle development. Retinol’s value, in that model, is not marketing tradition; it is its capacity to reduce stratum corneum stiffness, narrow the elastic gap, and restore the upper dermal soft layer, with type V collagen highlighted as a structural component of that functional softness that declines after 40 and can be restored ex vivo within two days. In other words, the future conversation is no longer what ingredient works, but what intervention restores the mechanical architecture that prevents deepening of biological damage.
This naturally leads to a broader regenerative thesis: the extracellular matrix is not a passive scaffold; it is a living biomechanical signalling system, and fibroblasts are not simply collagen factories; they are decision makers responding to tension, inflammation, oxidative stress, and nutrient state. When the extracellular matrix fragments, through matrix metalloproteinases, glycation crosslinks, oxidative damage, or senescence driven inflammatory signalling, fibroblasts lose their normal mechanotransductive cues and drift toward dysfunction. Modern biostimulation is, at its best, an attempt to re-establish the biological conditions that allow fibroblasts to behave like competent fibroblasts again: to build organized collagen, maintain elastin integrity, and sustain dermal hydration and viscoelasticity. That is why the regenerative aesthetics trends talk resonated with what was repeatedly emphasized throughout the congress as the future: post COVID patients are prioritizing tissue quality and natural outcomes, and the core regenerative mechanisms described, fibroblast activation, controlled inflammatory response, neoangiogenesis, immune modulation, extracellular matrix remodelling, and increased collagen and elastin synthesis, are precisely the biological levers that determine a natural, durable result. The message is not anti-filler in an ideological sense; it is a strategic re balancing where correction is no longer the primary endpoint; tissue biology is.
The longevity framing gained even more weight when perceived age was linked to systemic health. The congress content described large cohort work showing that those who look younger tend to have lower prevalence of multiple age associated conditions and better cognition, and that looking older does not correspond to fewer diseases. This is consistent with prospective population evidence showing perceived age is associated with morbidity and mortality, supporting the concept that facial aging can function as a biomarker of biological aging, not merely a cosmetic characteristic. This is clinically relevant because it elevates skin longevity from aesthetic preference to biological signal: the face becomes a readout of systemic resilience, inflammatory burden, and tissue integrity.
From a future looking perspective, IMCAS also made clear that the next decade will be shaped by integration of intercellular signalling therapies, cellular housekeeping biology, and senescence modulation. Exosomes and extracellular vesicles are biologically compelling because they constitute a native communication language between cells, carrying proteins, lipids, and nucleic acids capable of influencing inflammation, angiogenesis, fibroblast behaviour, and wound repair. Reviews summarizing translational and early clinical work support biological plausibility, but they also highlight current limitations: variability in vesicle source, lack of standardized dosing and potency assays, heterogeneity of manufacturing, and insufficient large controlled clinical trials for many marketed claims. There are also public scrutiny and regulatory concern around unapproved exosome products marketed in aesthetic settings, reflecting the reality that a biologically active product category will demand medical grade standardization and oversight if it is to become credible and widely adopted. What to expect, therefore, is not a simple exosome expansion, but a separation between rigorously manufactured, clinically studied extracellular vesicle therapies and non-standardised commercial offerings that may face increasing restriction.
Proteostasis and autophagy emerged as essential concepts for skin longevity because durability is not only about collagen synthesis; it is also about cellular quality control. Autophagy is the system by which cells clear damaged proteins and organelles, including dysfunctional mitochondria. Impairment of autophagic flux contributes to accumulation of oxidative damage, metabolic inefficiency, and reduced regenerative capacity, affecting keratinocytes and fibroblasts alike. Dermatologic research increasingly emphasizes that autophagy intersects with oxidative stress, senescence, and tissue repair, and that its decline is a hallmark mechanism of aging in skin biology. The practical implication is clear: biostimulation in a metabolically compromised environment produces less predictable outcomes. Regeneration becomes more durable when cellular maintenance systems are supported, redox balance is improved, and mitochondrial function is preserved.
Senescence modulation is the most direct bridge between longevity science and clinical dermatology. Senescent cells accumulate with age and secrete SASP mediators that amplify inflammation and extracellular matrix degradation. The congress content explicitly referenced senomorphic and senolytic strategies as emerging approaches, alongside mitochondrial fitness and a behaviour targeting treatment philosophy rather than simple volume restoration. In the broader scientific landscape, topical geroscience approaches are no longer theoretical; they have entered human exploratory trials, including topical rapamycin studies reporting measurable changes in senescence associated markers and clinical parameters in skin, supporting proof of principle for topical modulation of aging biology. This is a significant signal: the next generation of antiaging is increasingly likely to be defined by quantifiable effects on senescence burden, inflammatory signalling, and structural preservation rather than by surface smoothing alone.
The clinical evidence base for collagen stimulation is evolving toward a more mature, trial driven framework. Prospective controlled studies and expanding systematic reviews support the efficacy and safety of biostimulatory agents such as poly L lactic acid and calcium hydroxylapatite for tissue quality and contour outcomes, with increasing attention on protocol variables that directly influence biological remodelling: dilution, depth, session spacing, and objective assessment of dermal change. Hyperdiluted calcium hydroxylapatite is particularly relevant to the longevity conversation because it shifts the modality away from volumisation and toward collagen stimulation and skin quality improvement, aligning it with regenerative goals. What is coming next is not simply wider adoption, but better protocol science and more robust endpoint selection, including ultrasound measures of dermal thickness, elastography, and validated clinical skin quality scales.
The genomics and metabolomics concepts captured at IMCAS align with how longevity medicine is developing globally: prediction combined with targeted correction. Genomics indicates predisposition while metabolomics reflects real time biochemical activity, allowing clinicians to connect inflammation, glycation pathways, oxidative stress, collagen breakdown, and antioxidant capacity with individualized interventions. This is clinically meaningful because two patients receiving the same biostimulatory protocol may generate different collagen outcomes depending on mitochondrial competence, oxidative stress burden, metabolic status, barrier dysfunction, and inflammatory tone. Longevity focused dermatology therefore becomes a precision discipline, integrating biological state with procedural planning.
If I had to summarize what IMCAS 2026 felt like scientifically, it was this: regenerative aesthetics is no longer an isolated aesthetic trend; it is becoming the clinical expression of longevity biology. The emerging standard is an evidence-based plan that preserves extracellular matrix architecture, protects mitochondria and proteostasis, reduces inflammaging and senescence burden, stabilizes barrier and microbiome ecology, and uses biostimulatory tools, topical, injectable, device based, and lifestyle linked, not to change the face, but to restore tissue behaviour. That is why natural results are dominating: not because patients want less medicine, but because advanced medicine now aims to make tissue function better, not simply look different.
What to expect from the future, based on what was visible at IMCAS, is a move toward structured longevity protocols with defined biological endpoints. We will see more integrated sequencing where barrier and microbiome management are treated as prerequisite steps, not adjuncts. We will see wider use of objective imaging and AI phenotyping to stratify aging patterns and predict responders. We will see a stronger evidence gap addressed in areas like exosome biology and senotherapeutics, with regulatory pressure forcing standardization and clinical trials. We will see more emphasis on mitochondrial support and proteostasis as necessary foundations for durable regeneration. Above all, we will see that the most credible antiaging will be the one that proves biological durability.
From a practical clinical standpoint, this longevity model translates into a different way of selecting patients, planning sequencing, and defining success. The first step is to identify whether the primary limitation is structural extracellular matrix collapse, inflammatory load, barrier fragility, metabolic compromise, or a combination, because each of these conditions changes how the tissue responds to stimulation. In early aging, where fibroblast capacity remains relatively intact, regenerative interventions can be preventive and lighter, focusing on maintaining dermal quality and preventing mechanical imbalance. In moderate aging, where the extracellular matrix is fragmented and inflammation is higher, sequencing becomes critical: barrier repair and inflammation control precede strong stimulation, because stimulating a fibroblast in an inflamed, oxidatively stressed environment risks unpredictable remodelling. In advanced aging, where senescence burden and glycation stiffness dominate, realistic expectations and combination strategies become essential, and the value of senomorphic modulation and metabolic stabilization becomes more relevant.
Success, in this framework, should be measured as longevity outcomes rather than only cosmetic change. That includes improved skin resilience, improved structural coherence, and durable quality improvement over months rather than transient correction over days. It includes a shift toward objective assessment where feasible, incorporating imaging metrics such as dermal thickness, tissue stiffness patterns, and changes in skin quality parameters. The role of the dermatologist expands: not only to inject or treat, but to curate a biologically coherent plan that preserves function and extends healthspan.
Bioderma’s relevance within this longevity-oriented model is direct and scientifically coherent because the first layer of skin longevity is barrier competence and inflammatory control. When the barrier is impaired, transepidermal water loss increases, microbial balance destabilizes, and low-grade inflammatory signalling intensifies. That environment accelerates extracellular matrix fragmentation through increased oxidative stress and protease activity, and it reduces the predictability of any regenerative intervention because fibroblasts respond poorly when chronic inflammation, barrier disruption, and metabolic stress coexist. A barrier first strategy therefore becomes foundational, not supplementary. The practical consequence is that medical grade dermocosmetic support is no longer positioned as maintenance after procedures only, but as part of the core longevity protocol that creates the biological conditions required for durable tissue quality improvement. Within this approach, formulations that support lipid restoration, reduce irritant driven inflammation, and respect microbiome equilibrium can be framed as protective measures for dermal structure over time, because they reduce the upstream triggers that amplify inflammaging and accelerate matrix degradation. In a longevity perspective, the most credible skin care is the one that improves functional resilience, reduces inflammatory reactivity, and supports long term stability.
This aligns with what was consistently emphasized at IMCAS, namely that the future of aesthetic dermatology is built on prevention, preservation, and biological coherence, with natural outcomes emerging as a clinical consequence of restoring tissue physiology rather than imposing external correction.
Report written by Dr Lev Naidoo, (Dermatologist, South Africa)
Chairs : Dr Geisa Costa and Dr Edward Lain
Speakers: Dr Mukta Sachdev (India) and Dr Elodie Valin (France)
Beauty that lasts- The longevity aesthetic blueprint
Speaker: Dr Mukta Sachdev (India)
Introduction to Practical Longevity
Dr Sachdev introduced longevity dermatology as a shift in the aesthetic focus realigning toward supporting the biological youth of the skin for longer, rather than chasing the appearance of looking younger. This set the tone for IMCAS 2026, as it guided the clinician toward fostering a more holistic approach to our patients’ health. The presentation was an integration of epigenetics, nutricosmetics and lifestyle measures to support the skin’s longevity.
The Skin is the frontline longevity organ
The move in focus sees a departure from anti-aging toward supporting the health span of the skin by addressing the skin’s structure, function and resilience by investing in the skin barrier, immunological balance and preservation of extra cellular matrix quality. Dr Sachdev highlighted that present approaches must also address stem cell failure and chronic inflammaging which accelerate skin decline.
Aesthetic Dermatology Innovations
New aesthetic treatments should:
Tips for Longevity in Aesthetics
Redefining Treatment Goals
Dr Sachdev concluded that by approaching the health of the skin comprehensively, goals of skin longevity including skin resilience and durability with a long- term view toward tissue repair and natural outcomes could be attained more effectively.
Proteome Protection as the key to skin cellular longevity- A new topical approach inspired by recent discoveries in age-related diseases
Speaker: Dr Elodie Valin (France)
Ecobiological Approach
Dr Elodie Valin , the NAOS scientific director, presented on the concept of proteome protection and its significance in cellular longevity.
Ecobiology combines ecosystem science with biology, treating the skin as a living ecosystem that interacts with the environment, with a goal of supporting the skin’s biology for long-lasting benefits, rather than merely treating signs of aging.

Research Background
The NAOS partnership with world- renowned biologist Professor Radman, provided direction for a change in focus from the genome to the proteome when approaching skin health. Professor Radman’s research demonstrated how extremophilic bacteria were able to survive in the most challenging environments, through the bacteria’s capability to repair damaged DNA via their proteomic make-up that included inoxidable repairing proteins.

This is research was then harnessed not just for longevity, but for a better quality of life.
The Proteome and the Skin

The proteome is the complete set of proteins in a cell or tissue. Proteins are the most abundant component in the body after water and as such are of key importance in supporting skin longevity through their dual structural and functional roles.
Protein Carbonylation in the Skin

Protein carbonylation is the one type of oxidative damage that is specific to proteins. It freezes a protein into a three-dimensional structure that is non- functional, which in time aggregates into larger structures that are toxic to our cells, making them both a marker and accelerator of cell aging.
Clinically protein carbonylation in the skin presents with epidermal dehydration, loss of radiance, a decrease in dermal density and rhytid appearance.
Proteome Protection and Skin Aging
The exobiological approach of protecting the proteome therefore protects from clinical signs of aging.

This is achieved via the patented biotechnology , extracted from Arthrobacter agillis, containing chaperone-like properties, meaning it can physically shield the protein molecule and antioxidant properties.

Clinical Proof
A hemi-face 6-month clinical study demonstrated this patented proteome technology’s superior result when used in a serum with a neutral cream in comparison to the sole use of the neutral cream in terms of skin firmness, density, evenness of complexion and rhytid improvement.
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chairs: Prof Martina J Kerscher and Dr Shadi Kouros
Speakers: Prof Hassan Galadari, Dr Shadi Kourosh and Dr Barbara Dalbos Bouillard
Reversing skin age: epigenetic science meets aesthetics
Speaker: Prof Hassan Galadari
Introduction to Epigenetics

Prof. Galadari introduced this lecture on epigenetics by reminding us that whilst our genetic makeup accounts for our diversity, the greater nuances reflected by the epigenetic read on our genes further extrapolates this diversity.
Key: All the cells in our body have the same genetic make-up. Epigenetics refers to the regulation of gene expression without altering the underlying DNA sequence. It represents the functional interface between the genome and the environment.
Mechanisms of Epigenetics
Epigenetic changes allow genes to be expressed in various ways.
Key mechanisms include:

Gene Activation and Suppression
Through evolving epigenetic science, we understand as individuals age, certain genes may become expressed or suppressed.
Extrinsic factors including UV exposure, toxin, smoke , pollution, stress, altered sleep patterns and suboptimal nutritional intake may turn protective genes off, compromising our overall health, including our skin’s health.
Protective Factors
Prof. Galadari went on to highlight that this process may also be positively modified in the skin by the inclusion of key skin practices that turn on reparative gene pathways i.e.: supportive epigenetic pathways.

Clinical Procedures and Epigenetics
Prof. Galadari further highlighted the synergism of these protective skin ingredients with appropriate clinical procedures including laser and intense pulsed light treatments that also have the capacity to positively affect our epigenetic read.

Chang et al. J Invest Derm.2013
Hamelin MR. Photomed Laser Surg. 2018
Haykal D et al. J Cosmet Dermat. 2025
Epigenetics in Precision Skincare
Speaker: Dr Shadi Kourosh
Introduction
Dr Adrianne Shadi Kourosh’s lecture was a continuation into how we can practically utilize principles of epigenetics in our clinics. She opened with key supportive skincare and procedures we can include to switch on protective epigenetic reads and further went on to explain the importance of timing select skincare ingredients to the natural circadian rhythm of the skin.
The key principles outlined included:
The Skin’s Circadian Clock
Dr Kourosh further explained the skin has its own circadian clock and skincare should support this synergistically by protecting our DNA during the day and feeding skin renewal at night. She highlighted that by timing our skincare we can potentiate its clinical benefits.

Epigenetics and hyperpigmentation: how can epigenetics go beyond the classic “anti-tyrosinase” approach?
Speaker: Dr Barbara Dalbos Bouillard
Introduction to hyperpigmentation
Hyperpigmentation is often viewed as an excess of pigment produced by melanocytes following exposure to acute inciting stimuli. Dr Bouillard challenged this perspective, postulating that it may instead be an alteration in melanocyte behaviour with immunological memory cells driving the process of pigment production even beyond the point that the inciting stimulus has passed .

Epigenetic factors in hyperpigmentation
Exposure to inflammation modulates the expression of key melanocyte regulators. This modulation affects both the synthesis of pigment and the skin’s sensitivity to pigment-promoting stimuli.

Key: The skin can remain affected long after an initial melanin-inducing trigger has disappeared.
Concept of cellular memory

Hyperpigmentation is initiated by exposure and inflammation but can become self- perpetuating even when the exposing stimuli or inflammatory insult is no longer present. Cellular memory may therefore result in the persistent upregulation of melanogenic pathways, in the absence of ongoing stimuli.
Clinical Observations
The process of cellular memory explains how pigmentation can persist for months or even years without ongoing visible inflammation, suggesting a long-lasting functional programming of melanocytes instead of just temporary activation.
This impact may be more intense in the higher Fitzpatrick skin types, where inflammatory responses lead to greater pigment production, with a slower reset a slower to baseline tone.

Treatment challenges and importance of upstream intervention
Many treatments fail as they have focused on decreasing pigment production rather than subduing persistent inflammatory memory. Strategies must focus upstream on controlling inflammation. The prevention and control of cellular memory is crucial for durable results in the context of hyperpigmentation.
Dr Bouillard highlighted that topical steroid fails as they only temporarily decrease inflammation but do not have the capacity to act on important chronic melanogenic gene promotors like MITF.

She proposed consideration of ingredients like tranexamic acid that have both capacity to decrease inflammation and regulate MITF activity.
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Speaker : Dr Seemal Desai
Oral therapies for melasma
Speaker: Dr Seemal Desai
Introduction
Dr Desai introduced the topic by establishing that melasma cannot be addressed by monotherapy. In addition to topical treatment modalities, he highlighted five oral treatments that may be incorporated in the treatment of melasma.
Tyrocutin
Oral tyrocutin (coumaric acid) is the latest oral ingredient introduced for melasma at this congress. It binds directly to tyrosinase and TRP-2 inhibiting melanin synthesis as well as having an anti-inflammatory effect.
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chairs: Dr Foteini Bageorgou and Dr Marina Landau
Speaker: Dr Mukta Sachdev
Peels for Skin of Colour- Update and Newer Formulations in 2026
Speaker: Dr Mukta Sachdev
Key Trends for Skin of Colour in 2026 : Superficial peels are preferred for their safety and efficacy in skin of colour patients. There is a shift toward gentler, lower concentration peels that minimise the risk of both post-inflammatory hyperpigmentation and downtime.
Priming is a Key Step : The priming process should be respected for two to four weeks prior to the procedure. Pre-peel, it allows for detection of potential causes for clinical intolerance and in so doing decreases the risks of adverse effects. It also allows time to establish whether a patient can be compliant with a skincare routine. Priming further facilitates uniform penetration of the peeling ingredient during the peel and allows for a smoother healing process following the peel.
Priming Agents : These may include sunscreen, topical alpha- or beta- hydroxy acids, retinoids or ingredients that subdue the melanogenic pathways.
Innovative Delivery Systems : These include buffered acids, polymer-controlled penetration and lipophilic trichloroacetic acid preparations which deliver consistent, controlled penetration and reduce the unpredictability of depth penetration.
Home Care : Finally, Dr Sachdev touched on the importance of home care instructions to ensure the patient is aware of how to protect and support the skin following the chemical peel.
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chairs: Dr Ferial Fabian and Dr Michael H Gold
Speaker: Dr Ross Raduscky
Beyond the scale: the nutritional pitfalls of diet extremes and GLP-1s
Speaker: Dr Ross Raduscky
The Ozempic Face Phenomenon
Ozempic face is a colloquial term, that can be used to describe rapid facial deflation and increased skin laxity resulting in accelerated visible aging, hair shedding and slower post- procedural recovery following the use of GLP-1 agonists or rapid weight loss from extreme dieting. Counselling should occur at the beginning of diet extremes or GLP-1, therapy not after symptoms arise.
Nutritional Implications
Rapid intake reduction leads to loss of essential nutrients. This is common in GLP-1 patients who experience early satiety.
Decreased protein intake leads to decreased collagen synthesis and decreased lean muscle support.
Decreased essential fatty acid intake leads to a compromise of the lipid barrier and increased trans-epidermal water loss.
Defining Skin Aging End Points
Strategies for Protein Intake
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chairs: Dr Shepard Bhanon Khan, Dr Lucia Hue Fontaine and Dr Luiz Eduardo Toledo Avelar
Speaker: Dr Roger Chen
From GLP-1 to dual and triple agonists: Unveiling the efficacy of the uncertain hormones in weight management and metabolic disease
Speaker: Dr Roger Chen
Impact of Obesity:

Obesity affects multiple organs in the body including :
Shama Obes Rev 2010, 11; 808. Gun et al BMC Public Health 2009, 9: 88. Luppino
et al Arch Gen Psychiatry 2010, 67: 220-9
Historical context of obesity treatment
Previous treatment options showed limited success (5 to 8% weight loss.)
Surgical interventions resulted in significant weight loss but had concerning potential adverse effects (e.g. vitamin deficiencies, hypoglycaemia, hypotension.)
Introduction of incretin hormones GLP-1 and GIP


Combined effects of GLP-1 and GIP, leading to significant weight loss and improved glucose levels. They act on both receptors enhancing efficacy compared to previous treatments.
Mechanism of Action
Reduces hunger, increases satiety, causes delayed gastric emptying, improves glucose levels, and promotes weight loss.

Effects of GIP
Advances in anti-obesity medication Historical treatments
Old medications (e.g. Orlistat, contrave) provided minimal weight loss Liraglutide (once daily GLP-1) led to 8 to 10% weight loss.
New developments
Once weekly GLP-1 medication (e.g. Ozempic) can result in 14 to 16% weight loss.

Upcoming tri- molecule medication ( Retatrutide) targets multiple receptors, with phase 2 clinical results showing more upward of 20% weight loss.
Summary
Benefits of weight loss following pharmacotherapy :
1. Cardiovascular outcomes in type 2 diabetes
2. Cardiovascular outcomes in obesity
3. Heart failure (preserved ejection fraction)
4. Sleep apnoea
5. Fatty liver disease
6. Osteoarthritis
Update on the Ozempic face; causes, concerns and cures
Speaker: Dr Steven Dayan
New FDA approved indications for GLP treatments :
Can microdosing offer benefits?
Micro-dosing has still shown benefits of weight loss and a decrease in inflammatory markers for both GLP-1 and GIP agonists.
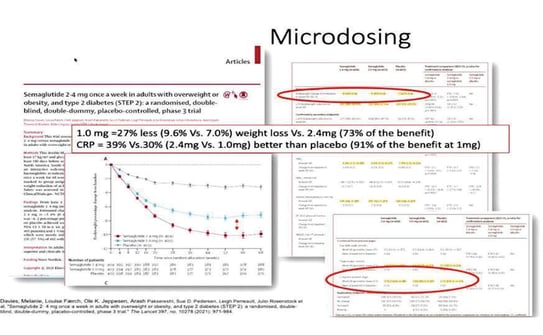
What’s new?
Phase 2 obesity trial NEJM 2023- 48 weeks led to 22.8 to 24.2% weight loss; approval expected 2026
New risks
Treatment options
Nikolis A, Dayanz S et al. Consensus Statements on Managing Aesthetic Needs in Prescription Medication-Driven Weight Loss Patients: An International, Multidisciplinary Delphi Study. J Cosmet Dermatol. 2025 Apr;24
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chairs: Dr Anne Colona and Dr Ronald Feiner
Speaker: Dr Mukta Sachdev
Neurocosmetics
Speaker: Dr Mukta Sachdev
Introduction
The integration of neurocosmetics into skincare routines is gaining popularity, with a focus on products that promote both skin health and emotional well-being.
Neurocosmetics are bioactive topical products that contain ingredients designed to work at a neurological level. When these products activate skin receptors and affect nerve endings they have the capacity to modulate skin cell functions.
Skin-brain axis: the biology that matters for cosmetics
Sensory neurons in turn communicate with other neighbouring cells resulting in:
What do theses active ingredients do?
Neurocosmetics modulate nerve signals in the skin and neurotransmitters release beneficial effects including:
Key neurocosmetic ingredients
Take home message
Growth in the field of neurocosmetics parallels the expanding awareness of the importance of skin-mind and gut health axis, representing interesting treatments options focused on fostering of skin longevity.
Report written by Dr Lev Naidoo (Dermatologist, South Africa)
Chair: prof Jeremy Magalon
Speaker: Dr Sophie Menes
Exosome therapy: how to repair skin barriers
Speaker: Dr Sophie Menes
Introduction
The discovery of exosomes dates back to the 1960s, with their significance regarding involvement in cell communication only appreciated much later. The messaging pathways they influence depend on their source, with clinical efficacy further nuanced by the process through which they are isolated.
The small size of exosomes and their bi-lipid membrane facilitate their entry into skin cells.

Exosomes deliver bioactive molecules to skin cells, influencing processes including collagen production, inflammation regulation and wound healing. They modulate important signaling pathways including the MAPK and TGF-b pathways, which are essential for skin repair and regeneration.

Sources of Exosomes for Cosmetic Application :
1. Human- derived products- not allowed in Europe
2. Plants- fruits, seeds, pollen
3. Animals- venom, milk ( colostrum), , mollusc, ticks
4. Microbial- fungal, bacterial, parasitic
5. Humans- tumours, body fluids, cells
6. Feral- derived animal exosomes
Exosome Isolation Techniques
Several mechanisms exist to isolate exosomes including ultracentrifugation, ultrafiltration and microfluidic devices. These techniques vary in complexity, purity and efficiency without standardization in method established.
Properties of exosomes :
1. Angiogenesis
2. Regulation of inflammation
3. Collagen synthesis and anti-aging effects
The role of exosomes in inflammatory dermatoses
Cho et al. (2018, 2020) demonstrated that adipose-derived mesenchymal stem cell-derived exosomes (ASC-exos) effectively ameliorate atopic dermatitis (AD) symptoms in mouse models. These exosomes reduce serum IgE, eosinophils, and inflammatory cytokines (IL-4, IL-5, IL-13, TNF-a, IFN- y, IL17, TSLP), while promoting skin barrier restoration by inducing ceramide production.
Skin Therapy and Regeneration
Exosomes may play a role in skin aging (through activity of senescence as well as extracellular matrix stimulation.)

The role of exosomes in wound and scar healing are related to their role in promoting cellular proliferation, granulation tissue formation and re-epithelialisation.
Biological challenges
Whilst there are many publications demonstrating the benefits to exosomes use, a major limitation with this therapeutic modality is the difficulty in precisely targeting cell types, while limiting off-target bio- distribution. The presence of naturally incorporated genetic impurities with potential immunogenicity poses an additional concern.
A better understanding of exosome engineering, uptake, cargo release and clearance is therefore required




Bioderma Congress Reports IMCAS 2025